QUESTION IMAGE
Question
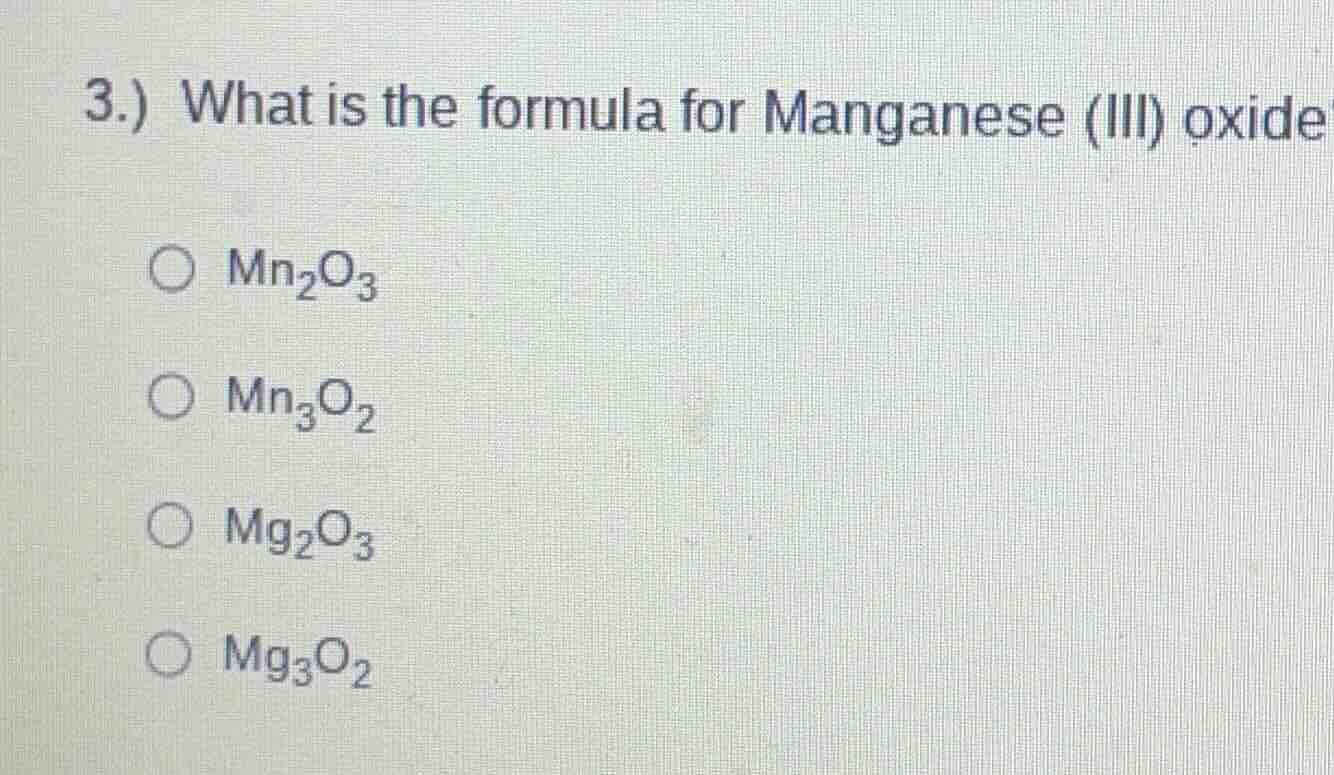
3.) what is the formula for manganese (iii) oxide
○ mn₂o₃
○ mn₃o₂
○ mg₂o₃
○ mg₃o₂
To determine the formula for Manganese (III) oxide, we first identify the ions. Manganese (III) has a charge of \( \text{Mn}^{3+} \) and oxide has a charge of \( \text{O}^{2-} \). Using the criss - cross method for ionic compounds (where the magnitude of the charge of one ion becomes the subscript of the other ion), we take the charge of Mn (\( 3^+ \)) as the subscript for O and the charge of O (\( 2^- \)) as the subscript for Mn. So the formula should be \( \text{Mn}_2\text{O}_3 \) (since 3 from Mn becomes the subscript of O and 2 from O becomes the subscript of Mn). Also, we know that the symbol for manganese is Mn (not Mg, which is magnesium), so we can eliminate the options with Mg.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \( \text{Mn}_2\text{O}_3 \)