QUESTION IMAGE
Question
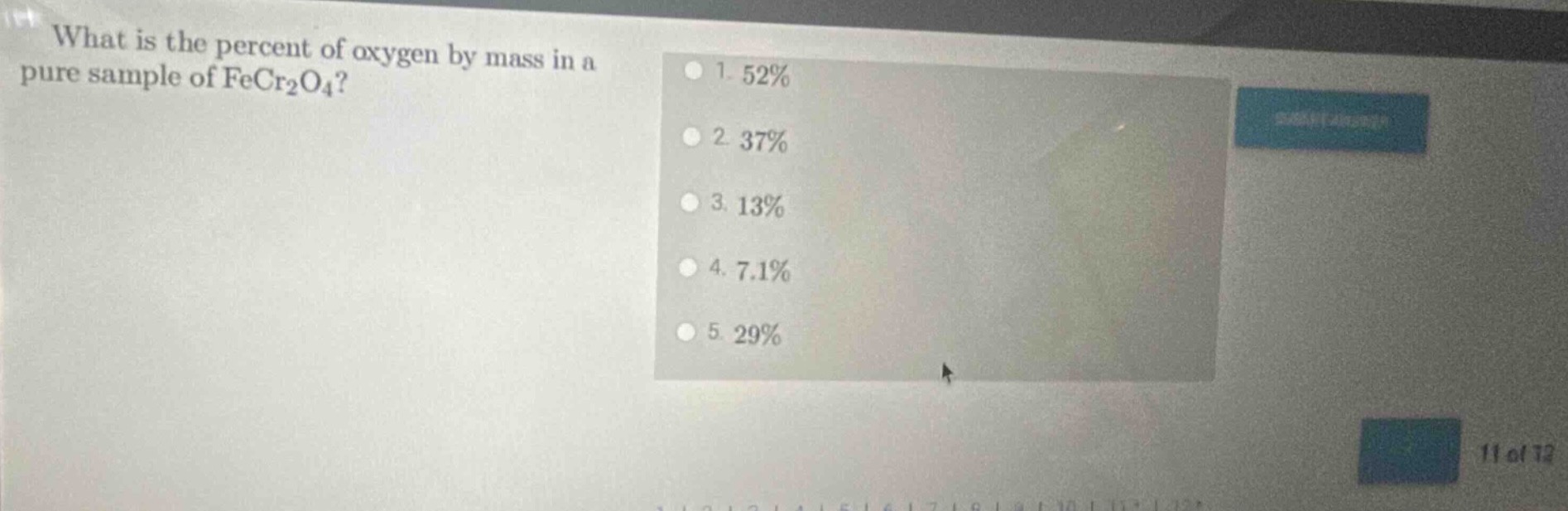
what is the percent of oxygen by mass in a pure sample of fecr₂o₄? 1. 52% 2. 37% 3. 13% 4. 7.1% 5. 29%
Step 1: Calculate molar mass of FeCr₂O₄
Molar mass of Fe: \( 55.85 \, \text{g/mol} \)
Molar mass of Cr: \( 52.00 \, \text{g/mol} \), so for \( 2 \, \text{Cr} \): \( 2 \times 52.00 = 104.00 \, \text{g/mol} \)
Molar mass of O: \( 16.00 \, \text{g/mol} \), so for \( 4 \, \text{O} \): \( 4 \times 16.00 = 64.00 \, \text{g/mol} \)
Total molar mass of \( \text{FeCr}_2\text{O}_4 \):
\( 55.85 + 104.00 + 64.00 = 223.85 \, \text{g/mol} \)
Step 2: Calculate mass percent of O
Mass percent of O = \( \frac{\text{Mass of O in compound}}{\text{Molar mass of compound}} \times 100\% \)
\( \frac{64.00}{223.85} \times 100\% \approx 28.6\% \approx 29\% \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- 29%