QUESTION IMAGE
Question
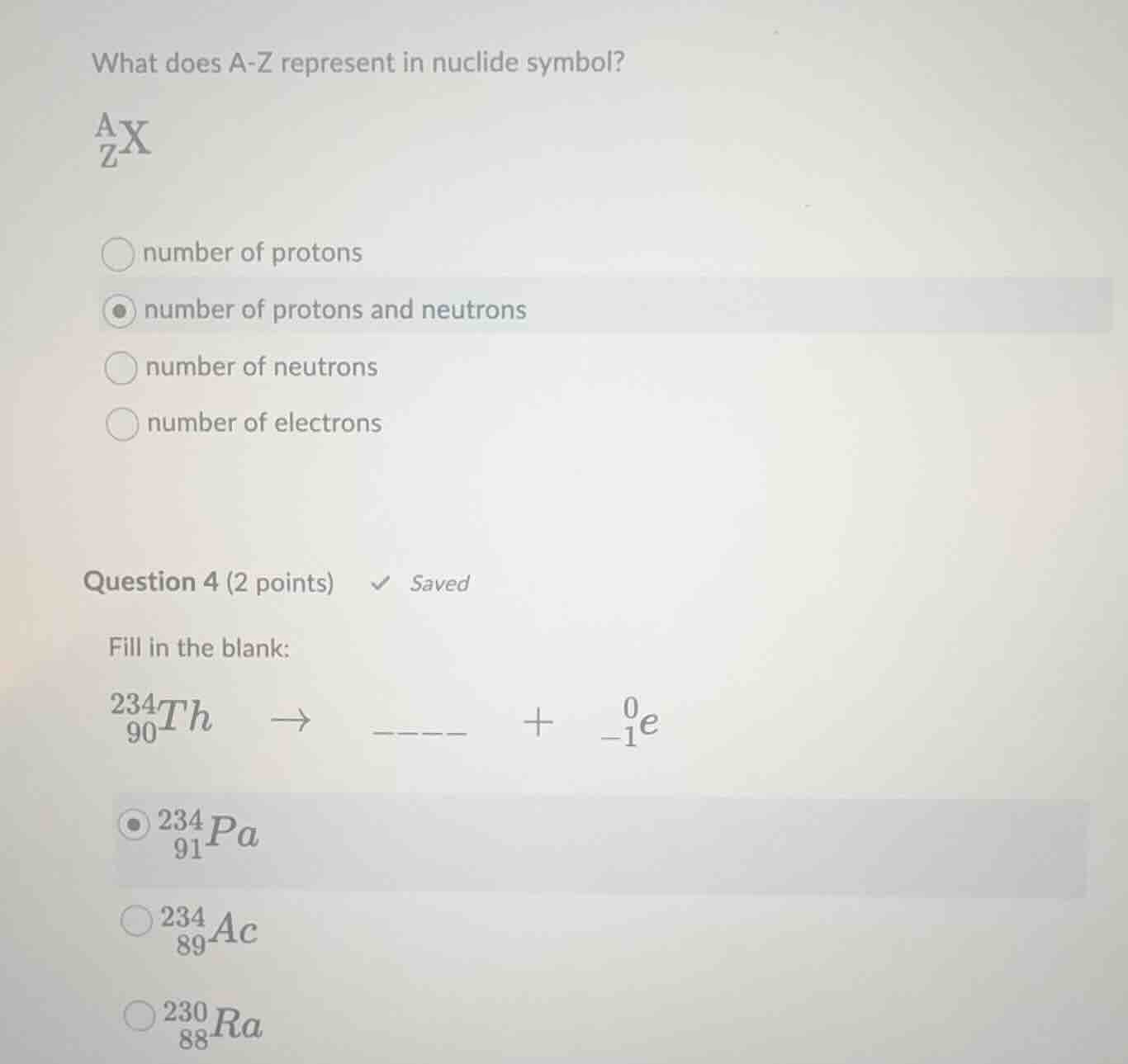
what does a - z represent in nuclide symbol?
\\(_{z}^{a}x\\)
\\(\bigcirc\\) number of protons
\\(\bigodot\\) number of protons and neutrons
\\(\bigcirc\\) number of neutrons
\\(\bigcirc\\) number of electrons
question 4 (2 points) \\(\checkmark\\) saved
fill in the blank:
\\(_{90}^{234}th\\ \\ \
ightarrow\\ \\ \\_\\_\\_\\_\\ \\ +\\ \\ _{-1}^{0}e\\)
\\(\bigodot\\) \\(_{91}^{234}pa\\)
\\(\bigcirc\\) \\(_{89}^{234}ac\\)
\\(\bigcirc\\) \\(_{88}^{230}ra\\)
First Question (What does A - Z represent in nuclide symbol?):
In the nuclide symbol \(\ce{_Z^A X}\), \(A\) is the mass number (number of protons + neutrons) and \(Z\) is the atomic number (number of protons). So \(A - Z\) is (number of protons + neutrons) - number of protons = number of neutrons. Wait, no, wait: Wait, the options: Let's re - check. Wait, the options are: number of protons (Z), number of protons and neutrons (A), number of neutrons (A - Z), number of electrons. Wait, the original question's options: Wait, the first option is "number of protons" (Z), second "number of protons and neutrons" (A), third "number of neutrons" (A - Z), fourth "number of electrons". So the correct answer for "What does A - Z represent" is the number of neutrons. Wait, but in the given image, the selected option was "number of protons and neutrons" which is wrong. Wait, no, let's re - do:
In nuclide notation \(\ce{_Z^A X}\):
- \(Z\): atomic number = number of protons (and also number of electrons in neutral atom).
- \(A\): mass number = number of protons + number of neutrons.
So \(A - Z=\) (number of protons + number of neutrons) - number of protons = number of neutrons. So the correct option is "number of neutrons".
Second Question (Fill in the blank: \(\ce{_{90}^{234}Th
ightarrow \_\_\_ + _{-1}^0 e}\)):
In a nuclear reaction, the sum of atomic numbers (subscripts) and mass numbers (superscripts) on both sides must be equal.
For mass numbers: On the left, mass number of Th is 234. On the right, mass number of the emitted particle (\(\ce{_{-1}^0 e}\)) is 0. So the mass number of the unknown nuclide (let's call it \(\ce{_y^x X}\)) must satisfy \(234=x + 0\), so \(x = 234\).
For atomic numbers: On the left, atomic number of Th is 90. On the right, atomic number of the emitted particle (\(\ce{_{-1}^0 e}\)) is - 1. So \(90=y+(- 1)\), so \(y=90 + 1=91\).
The element with atomic number 91 is Protactinium (Pa). So the nuclide is \(\ce{_{91}^{234}Pa}\), which matches the selected option in the image (even though the first question's selected option was wrong).
First Question Answer:
The correct option is: number of neutrons
Second Question Answer:
The correct option is: \(\boldsymbol{\ce{_{91}^{234}Pa}}\) (or in the option format: the option with \(\ce{_{91}^{234}Pa}\))
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
In a nuclear reaction, the sum of atomic numbers (subscripts) and mass numbers (superscripts) on both sides must be equal.
For mass numbers: On the left, mass number of Th is 234. On the right, mass number of the emitted particle (\(\ce{_{-1}^0 e}\)) is 0. So the mass number of the unknown nuclide (let's call it \(\ce{_y^x X}\)) must satisfy \(234=x + 0\), so \(x = 234\).
For atomic numbers: On the left, atomic number of Th is 90. On the right, atomic number of the emitted particle (\(\ce{_{-1}^0 e}\)) is - 1. So \(90=y+(- 1)\), so \(y=90 + 1=91\).
The element with atomic number 91 is Protactinium (Pa). So the nuclide is \(\ce{_{91}^{234}Pa}\), which matches the selected option in the image (even though the first question's selected option was wrong).
First Question Answer:
The correct option is: number of neutrons
Second Question Answer:
The correct option is: \(\boldsymbol{\ce{_{91}^{234}Pa}}\) (or in the option format: the option with \(\ce{_{91}^{234}Pa}\))