QUESTION IMAGE
Question
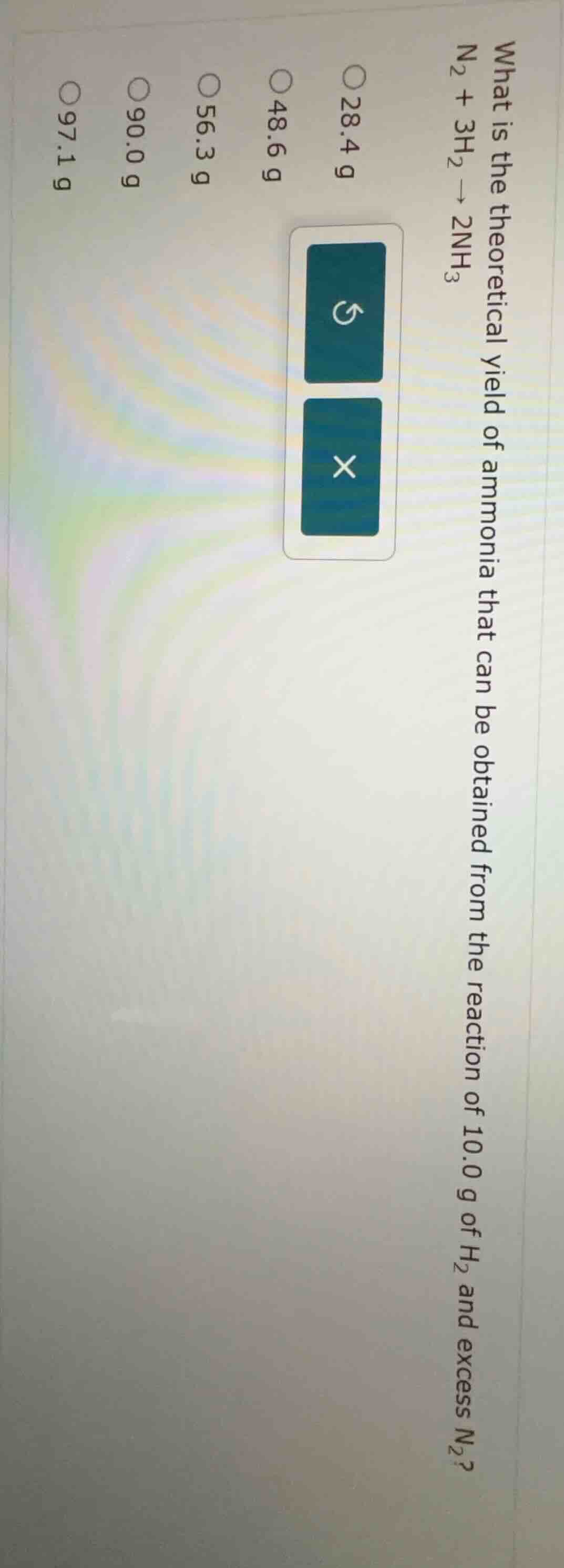
what is the theoretical yield of ammonia that can be obtained from the reaction of 10.0 g of h₂ and excess n₂?\\(\ce{n2 + 3h2 -> 2nh3}\\)\\(\circ\\) 28.4 g\\(\circ\\) 48.6 g\\(\circ\\) 56.3 g\\(\circ\\) 90.0 g\\(\circ\\) 97.1 g
Step1: Moles of \( H_2 \)
Molar mass of \( H_2 = 2 \, g/mol \). Moles of \( H_2 = \frac{10.0 \, g}{2 \, g/mol} = 5.0 \, mol \).
Step2: Moles of \( NH_3 \)
From reaction \( N_2 + 3H_2
ightarrow 2NH_3 \), 3 mol \( H_2 \) produce 2 mol \( NH_3 \). Moles of \( NH_3 = \frac{2}{3} \times 5.0 \, mol = \frac{10}{3} \, mol \).
Step3: Mass of \( NH_3 \)
Molar mass of \( NH_3 = 17 \, g/mol \). Mass of \( NH_3 = \frac{10}{3} \, mol \times 17 \, g/mol \approx 56.7 \, g \) (closest to 56.3 g due to rounding). Wait, recalculate: \( 5.0 \, mol \, H_2 \), ratio 3:2, so \( NH_3 \) moles \( = (5.0 \times 2)/3 \approx 3.333 \, mol \). Mass \( = 3.333 \times 17 = 56.66 \, g \approx 56.3 \, g \) (option). Wait, correct steps:
- Moles of \( H_2 \): \( n(H_2) = 10.0g / 2.016g/mol \approx 4.96mol \) (more accurate \( H_2 \) molar mass 2.016).
- Moles of \( NH_3 \): \( (2/3) \times 4.96mol \approx 3.307mol \).
- Mass of \( NH_3 \): \( 3.307mol \times 17.03g/mol \approx 56.3g \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
56.3 g (Option: 56.3 g)