QUESTION IMAGE
Question
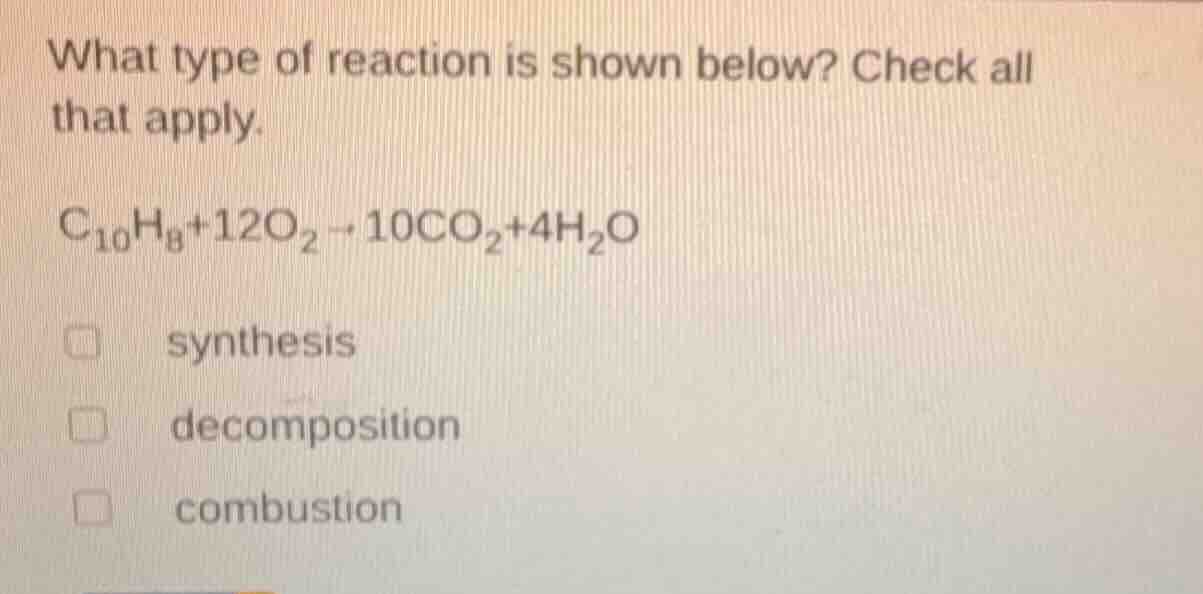
what type of reaction is shown below? check all that apply.
$\ce{c_{10}h_{8} + 12o_{2} -> 10co_{2} + 4h_{2}o}$
\\(\square\\) synthesis
\\(\square\\) decomposition
\\(\square\\) combustion
- Synthesis Reaction: A synthesis reaction is when two or more substances combine to form a single product (form: \( A + B
ightarrow AB \)). The given reaction has multiple products (\( CO_2 \) and \( H_2O \)), so it is not synthesis.
- Decomposition Reaction: A decomposition reaction is when a single compound breaks down into simpler substances (form: \( AB
ightarrow A + B \)). The given reaction has a reactant ( \( C_{10}H_8 \)) reacting with \( O_2 \), not a single compound breaking down, so it is not decomposition.
- Combustion Reaction: A combustion reaction involves a substance reacting with oxygen (\( O_2 \)) to produce carbon dioxide (\( CO_2 \)) and water (\( H_2O \)), often releasing energy. The reaction \( C_{10}H_8 + 12O_2
ightarrow 10CO_2 + 4H_2O \) fits this: a hydrocarbon (\( C_{10}H_8 \)) reacts with \( O_2 \) to form \( CO_2 \) and \( H_2O \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
combustion (the checkbox next to "combustion" should be selected)