QUESTION IMAGE
Question
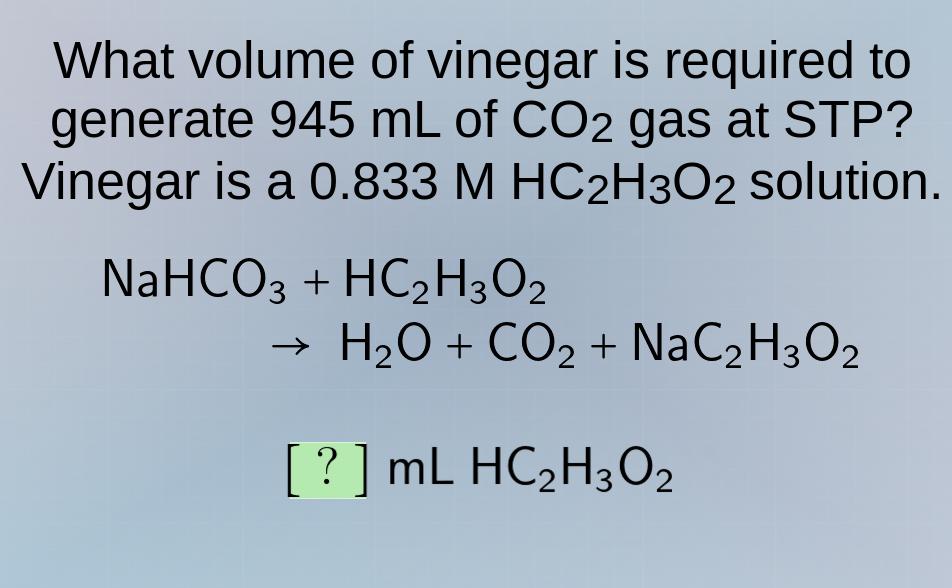
what volume of vinegar is required to generate 945 ml of co₂ gas at stp? vinegar is a 0.833 m hc₂h₃o₂ solution. nahco₃ + hc₂h₃o₂ → h₂o + co₂ + nac₂h₃o₂ ? ml hc₂h₃o₂
Step1: Find moles of \( CO_2 \) at STP
At STP, 1 mole of any gas occupies 22.4 L (or 22400 mL). So, moles of \( CO_2 \) = volume of \( CO_2 \) / molar volume at STP.
Volume of \( CO_2 = 945 \, mL = 0.945 \, L \)
Moles of \( CO_2 = \frac{0.945 \, L}{22.4 \, L/mol} \approx 0.04219 \, mol \)
Step2: Relate moles of \( CO_2 \) to moles of \( HC_2H_3O_2 \)
From the balanced equation \( NaHCO_3 + HC_2H_3O_2
ightarrow H_2O + CO_2 + NaC_2H_3O_2 \), the mole ratio of \( HC_2H_3O_2 \) to \( CO_2 \) is 1:1. So, moles of \( HC_2H_3O_2 = \) moles of \( CO_2 = 0.04219 \, mol \)
Step3: Find volume of \( HC_2H_3O_2 \) solution
Molarity (M) = moles of solute / volume of solution (in L). So, volume of solution (in L) = moles of solute / Molarity.
Molarity of \( HC_2H_3O_2 = 0.833 \, M \)
Volume of \( HC_2H_3O_2 \) solution (in L) = \( \frac{0.04219 \, mol}{0.833 \, mol/L} \approx 0.05065 \, L \)
Convert to mL: \( 0.05065 \, L \times 1000 \, mL/L = 50.7 \, mL \) (rounded to three significant figures)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 50.7 \) mL (or approximately 51 mL depending on rounding during steps)