QUESTION IMAGE
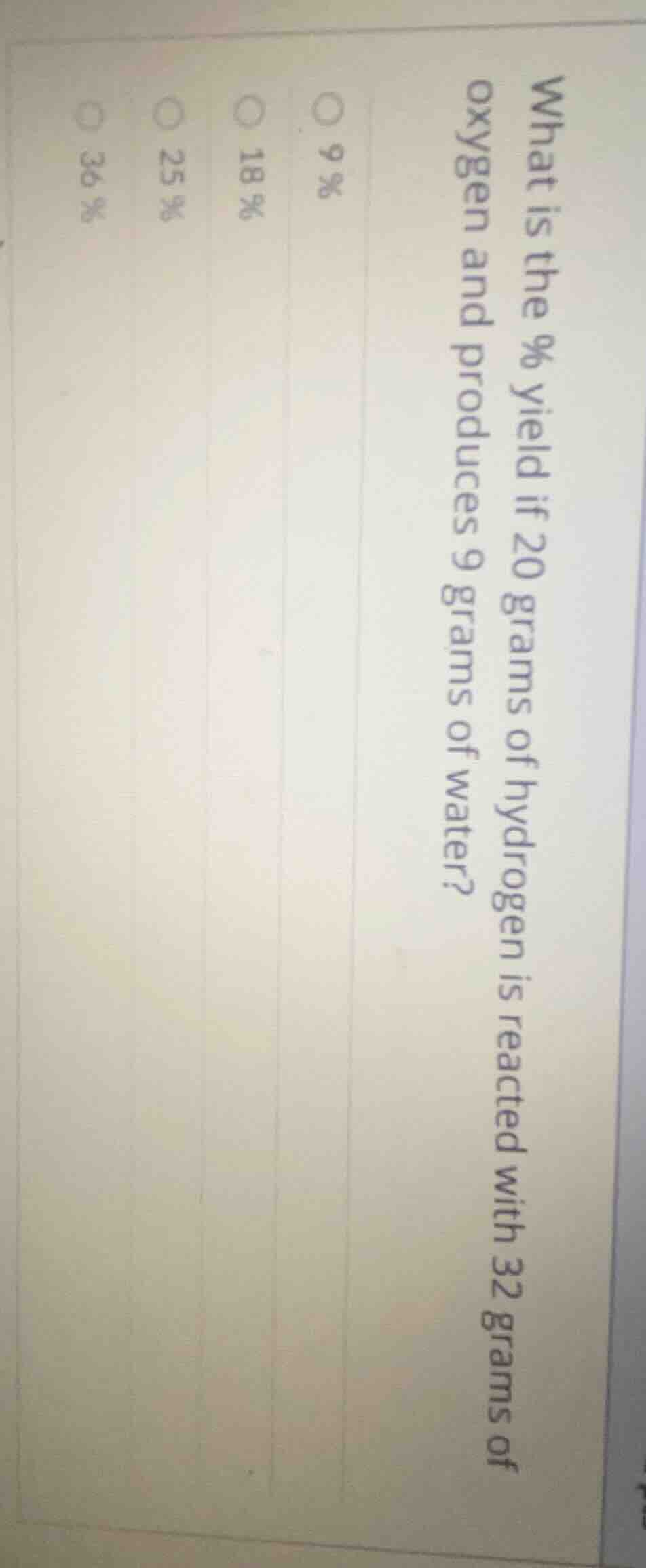
Question
what is the % yield if 20 grams of hydrogen is reacted with 32 grams of oxygen and produces 9 grams of water?
○ 9%
○ 18%
○ 25%
○ 36%
Step1: Write balanced reaction
$\ce{2H2 + O2 -> 2H2O}$
Step2: Find limiting reactant
Molar mass of $\ce{H2}=2\ \text{g/mol}$, moles of $\ce{H2}=\frac{20}{2}=10\ \text{mol}$
Molar mass of $\ce{O2}=32\ \text{g/mol}$, moles of $\ce{O2}=\frac{32}{32}=1\ \text{mol}$
From reaction, 1 mol $\ce{O2}$ reacts with 2 mol $\ce{H2}$. $\ce{O2}$ is limiting.
Step3: Calculate theoretical yield
1 mol $\ce{O2}$ produces 2 mol $\ce{H2O}$. Molar mass of $\ce{H2O}=18\ \text{g/mol}$
Theoretical yield $=2\times18=36\ \text{g}$
Step4: Compute percent yield
$\text{Percent Yield} = \frac{\text{Actual Yield}}{\text{Theoretical Yield}} \times 100\% = \frac{9}{36} \times 100\%$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
25 %