QUESTION IMAGE
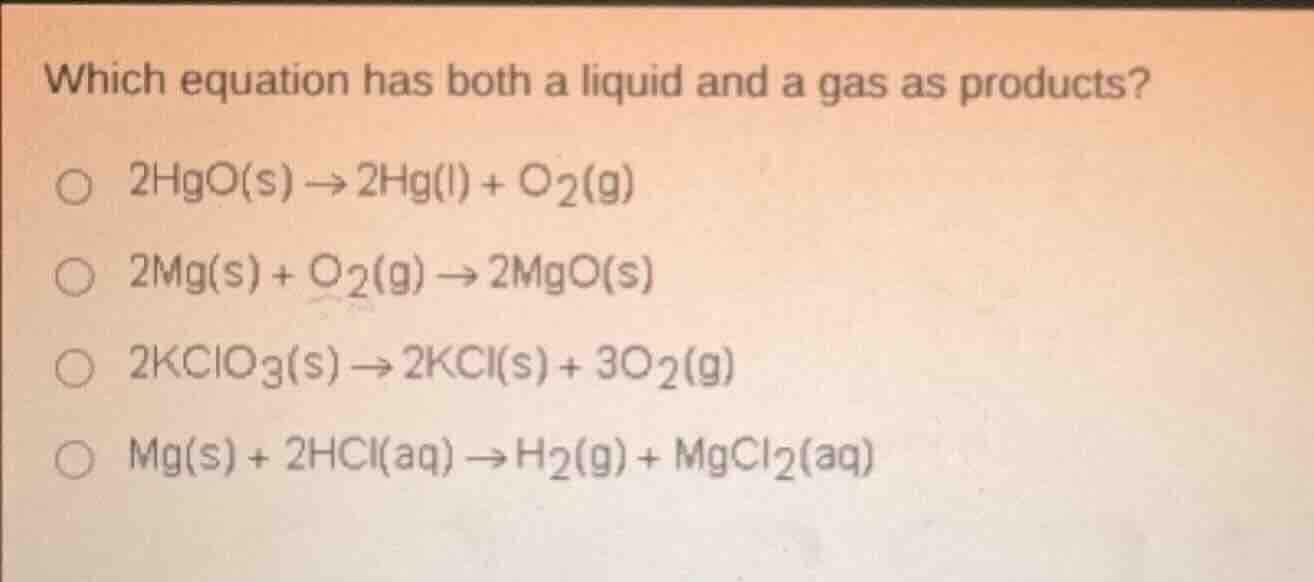
Question
which equation has both a liquid and a gas as products?
2hgo(s) → 2hg(l) + o₂(g)
2mg(s) + o₂(g) → 2mgo(s)
2kclo₃(s) → 2kcl(s) + 3o₂(g)
mg(s) + 2hcl(aq) → h₂(g) + mgcl₂(aq)
To determine which equation has both a liquid and a gas as products, we analyze each option:
- For \(2\text{HgO}(s)
ightarrow 2\text{Hg}(l) + \text{O}_2(g)\): The products are liquid mercury (\(\text{Hg}(l)\)) and gaseous oxygen (\(\text{O}_2(g)\)), so this has both a liquid and a gas.
- For \(2\text{Mg}(s) + \text{O}_2(g)
ightarrow 2\text{MgO}(s)\): The product is solid magnesium oxide, no liquid or gas (except reactant gas, but we look at products).
- For \(2\text{KClO}_3(s)
ightarrow 2\text{KCl}(s) + 3\text{O}_2(g)\): The products are solid potassium chloride and gaseous oxygen, no liquid.
- For \(\text{Mg}(s) + 2\text{HCl}(aq)
ightarrow \text{H}_2(g) + \text{MgCl}_2(aq)\): The products are gaseous hydrogen and aqueous magnesium chloride, no liquid (aqueous is a solution, not a pure liquid phase product in the sense of the question's likely intent, and the other product is aqueous, not liquid).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \(2\text{HgO}(s)
ightarrow 2\text{Hg}(l) + \text{O}_2(g)\)