QUESTION IMAGE
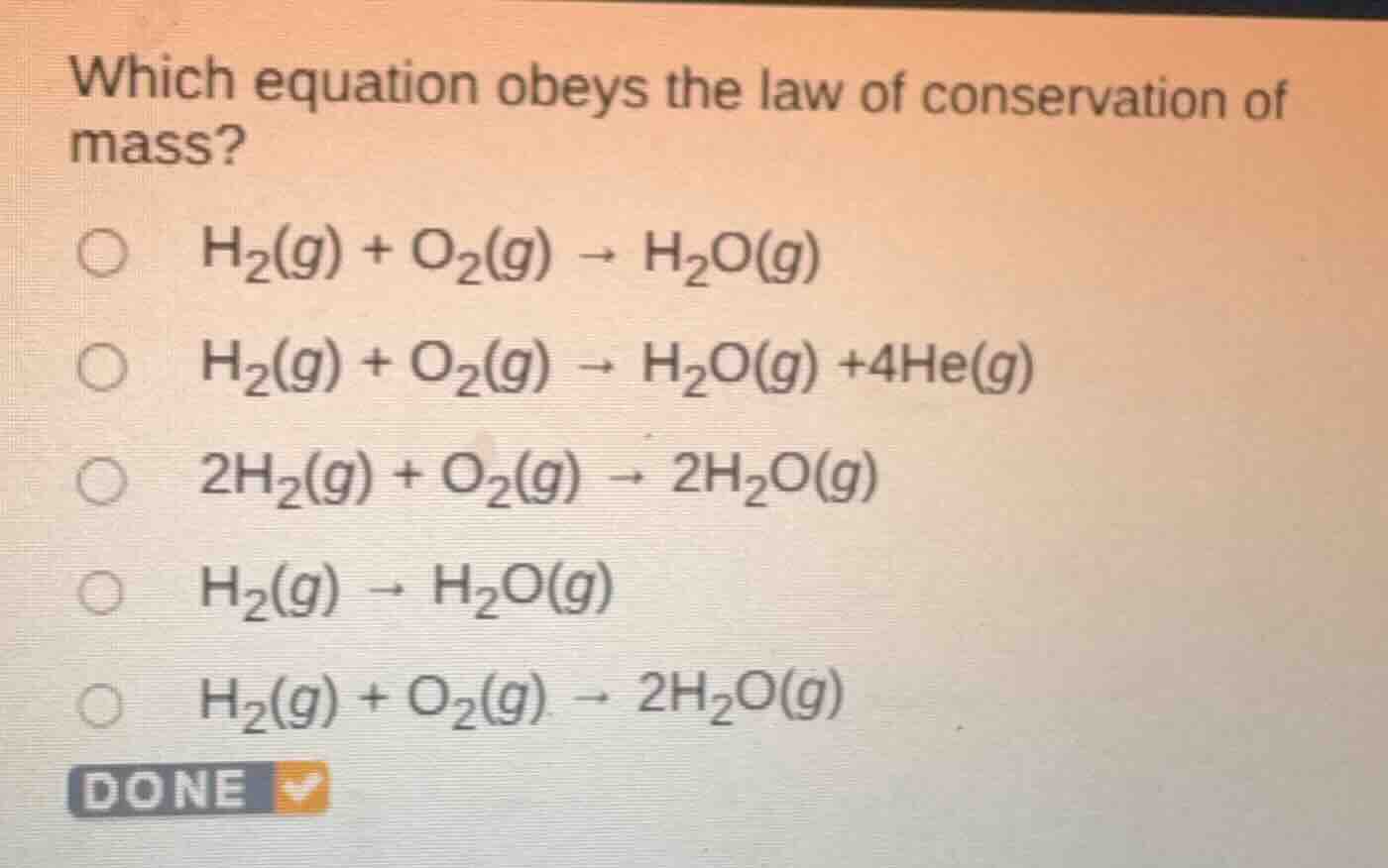
Question
which equation obeys the law of conservation of mass?
h₂(g) + o₂(g) → h₂o(g)
h₂(g) + o₂(g) → h₂o(g) +4he(g)
2h₂(g) + o₂(g) → 2h₂o(g)
h₂(g) → h₂o(g)
h₂(g) + o₂(g) → 2h₂o(g)
The law of conservation of mass requires that the number of each type of atom is the same on both sides of a chemical equation (balanced equation).
- For \( \ce{H2(g) + O2(g) -> H2O(g)} \): H: 2 vs 2; O: 2 vs 1 (unbalanced).
- For \( \ce{H2(g) + O2(g) -> H2O(g) + 4He(g)} \): New element (He) added, violates conservation (atoms not conserved from reactants).
- For \( \ce{2H2(g) + O2(g) -> 2H2O(g)} \): H: \( 2\times2 = 4 \) vs \( 2\times2 = 4 \); O: \( 2 \) vs \( 2\times1 = 2 \) (balanced).
- For \( \ce{H2(g) -> H2O(g)} \): O appears on product but not reactant (unbalanced, atoms not conserved).
- For \( \ce{H2(g) + O2(g) -> 2H2O(g)} \): H: 2 vs \( 2\times2 = 4 \); O: 2 vs \( 2\times1 = 2 \) (H unbalanced).
Only \( \ce{2H2(g) + O2(g) -> 2H2O(g)} \) has equal numbers of H and O atoms on both sides.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( \boldsymbol{2\ce{H2}(g) + \ce{O2}(g)
ightarrow 2\ce{H2O}(g)} \) (the third option: \( 2\ce{H2}(g) + \ce{O2}(g)
ightarrow 2\ce{H2O}(g) \))