QUESTION IMAGE
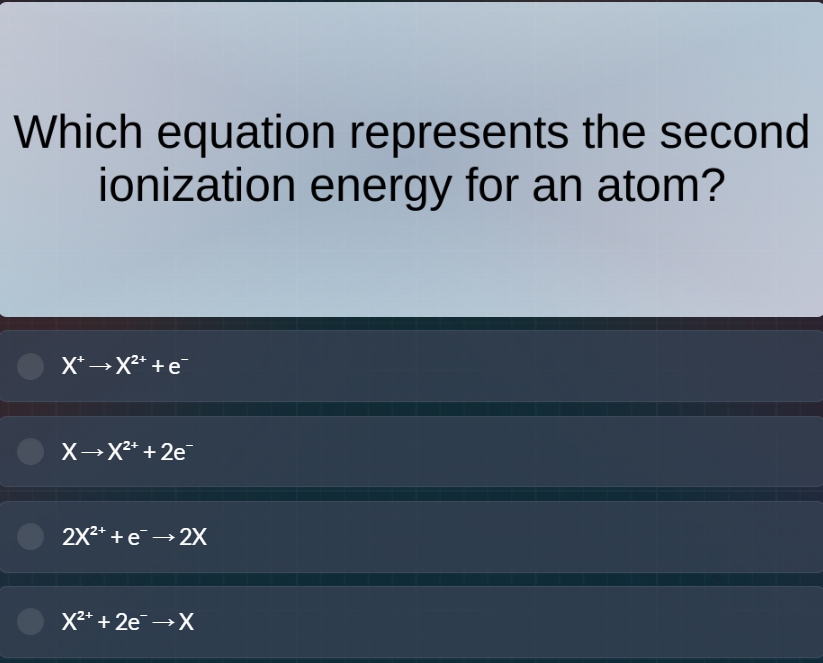
Question
which equation represents the second ionization energy for an atom?$x^+ \
ightarrow x^{2+} + e^-
$$x \
ightarrow x^{2+} + 2e^-$$
2x^{2+} + e^- \
ightarrow 2x$$x^{2+} + 2e^- \
ightarrow x$
Brief Explanations
Ionization energy is the energy required to remove an electron from a gaseous atom/ion. The second ionization energy specifically refers to removing one electron from a singly charged positive ion (formed after the first ionization) to create a doubly charged positive ion, releasing the electron.
- The first option shows a singly charged ion $X^+$ losing one electron to form $X^{2+}$, which matches the definition of second ionization energy.
- The second option shows a neutral atom losing two electrons, which is the sum of first and second ionization energies, not the second alone.
- The third and fourth options show electron gain (reduction), which is the opposite of ionization (oxidation/electron loss).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$X^+
ightarrow X^{2+} + e^-$