QUESTION IMAGE
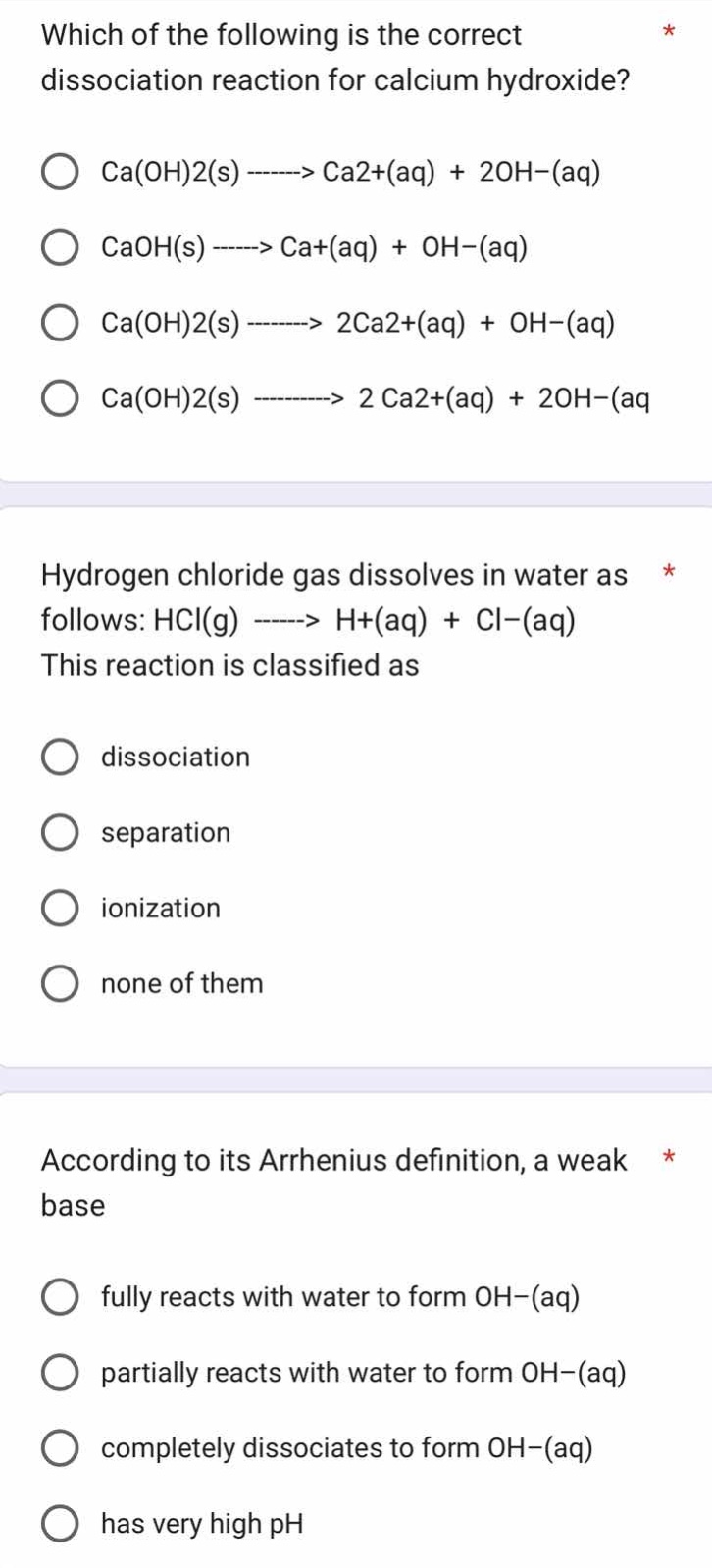
Question
which of the following is the correct
dissociation reaction for calcium hydroxide?
ca(oh)2(s) -------> ca2+(aq) + 2oh−(aq)
caoh(s) -----> ca+(aq) + oh−(aq)
ca(oh)2(s) -------> 2ca2+(aq) + oh−(aq)
ca(oh)2(s) ---------> 2 ca2+(aq) + 2oh−(aq
hydrogen chloride gas dissolves in water as
follows: hcl(g) -----> h+(aq) + cl−(aq)
this reaction is classified as
dissociation
separation
ionization
none of them
according to its arrhenius definition, a weak
base
fully reacts with water to form oh−(aq)
partially reacts with water to form oh−(aq)
completely dissociates to form oh−(aq)
has very high ph
- For calcium hydroxide dissociation: Calcium hydroxide has the formula $\text{Ca(OH)}_2$. It dissociates into one $\text{Ca}^{2+}$ ion and two $\text{OH}^-$ ions when dissolved in water, balancing charge and stoichiometry.
- For HCl reaction classification: Ionization is the process where a neutral molecule forms ions in solution. HCl is a neutral gas that forms $\text{H}^+$ and $\text{Cl}^-$ in water, which fits ionization (dissociation applies to ionic compounds breaking apart, not neutral molecules forming ions).
- For weak base definition (Arrhenius): An Arrhenius weak base does not fully react with water; only a portion of the base molecules produce $\text{OH}^-$ ions in solution, unlike strong bases that fully dissociate/react.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{Ca(OH)}2(\text{s})
ightarrow \text{Ca}2+(\text{aq}) + 2\text{OH}-(\text{aq})$
- ionization
- partially reacts with water to form $\text{OH}-(\text{aq})$