QUESTION IMAGE
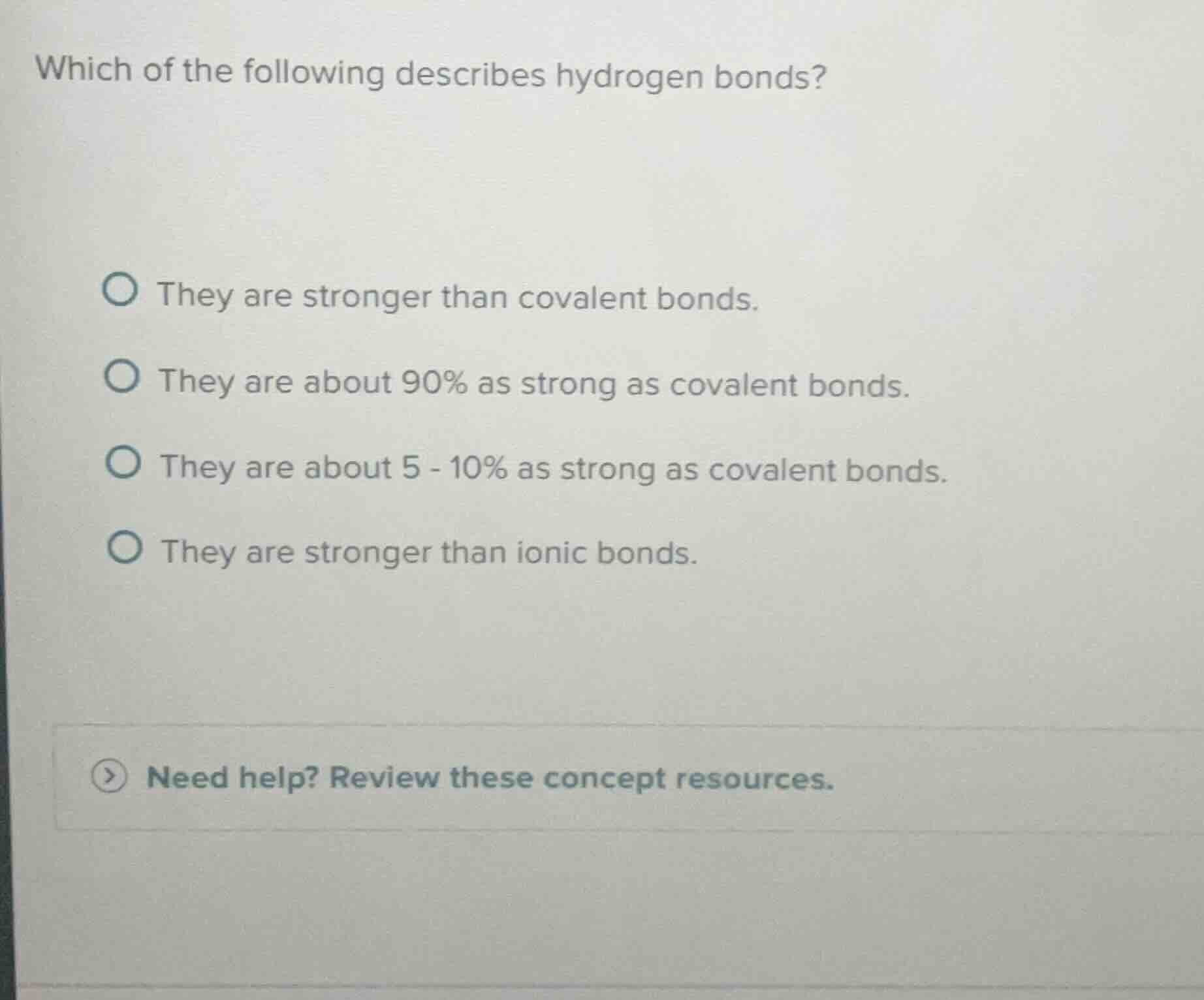
Question
which of the following describes hydrogen bonds?
they are stronger than covalent bonds.
they are about 90% as strong as covalent bonds.
they are about 5 - 10% as strong as covalent bonds.
they are stronger than ionic bonds.
need help? review these concept resources.
Brief Explanations
Hydrogen bonds are weak intermolecular forces, much weaker than intramolecular covalent and ionic bonds. Their strength typically falls in the range of 5-10% of the strength of a typical covalent bond.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
They are about 5 - 10% as strong as covalent bonds.