QUESTION IMAGE
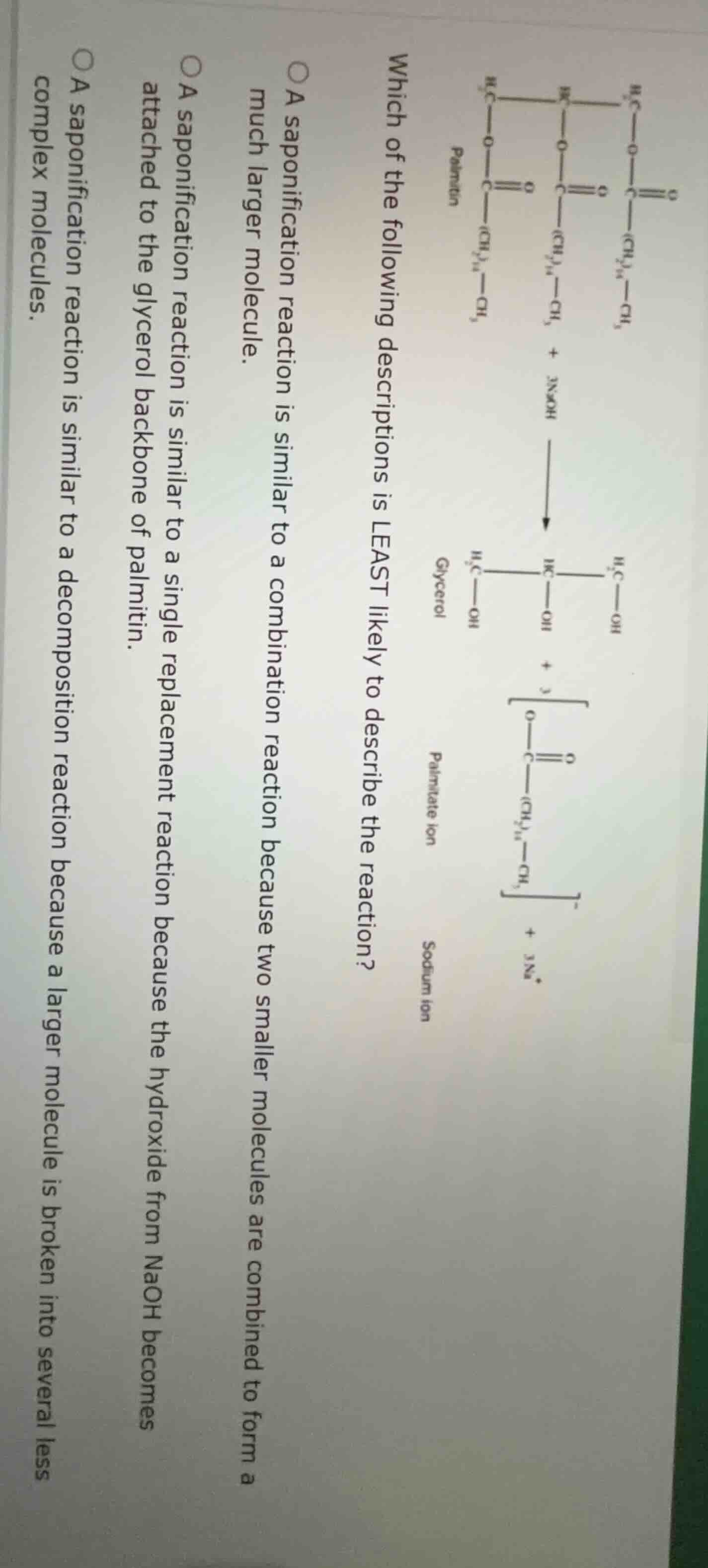
Question
which of the following descriptions is least likely to describe the reaction?
a saponification reaction is similar to a combination reaction because two smaller molecules are combined to form a much larger molecule.
a saponification reaction is similar to a single replacement reaction because the hydroxide from naoh becomes attached to the glycerol backbone of palmitin.
a saponification reaction is similar to a decomposition reaction because a larger molecule is broken into several less complex molecules.
- Option A: Combination reactions involve two or more substances combining to form a single product. Saponification is the hydrolysis of a fat (like palmitin) with a base (NaOH) to form glycerol and soap (palmitate + Na⁺), which is a decomposition (breaking a large molecule into smaller ones), not combination. So this description is incorrect.
- Option B: In saponification, the -OH from NaOH replaces the fatty acid chain attached to the glycerol backbone (ester hydrolysis with base), so it is similar to a single replacement (specifically, a hydrolysis - type replacement) as the hydroxide group replaces the ester - linked fatty acid. This is a valid description.
- Option C: Saponification breaks a large fat molecule (palmitin) into glycerol and soap molecules (smaller), which is similar to a decomposition reaction (a compound breaking into simpler substances). This is a valid description.
Since we need the LEAST likely description, Option A is the one that misrepresents the reaction type.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. A saponification reaction is similar to a combination reaction because two smaller molecules are combined to form a much larger molecule.