QUESTION IMAGE
Question
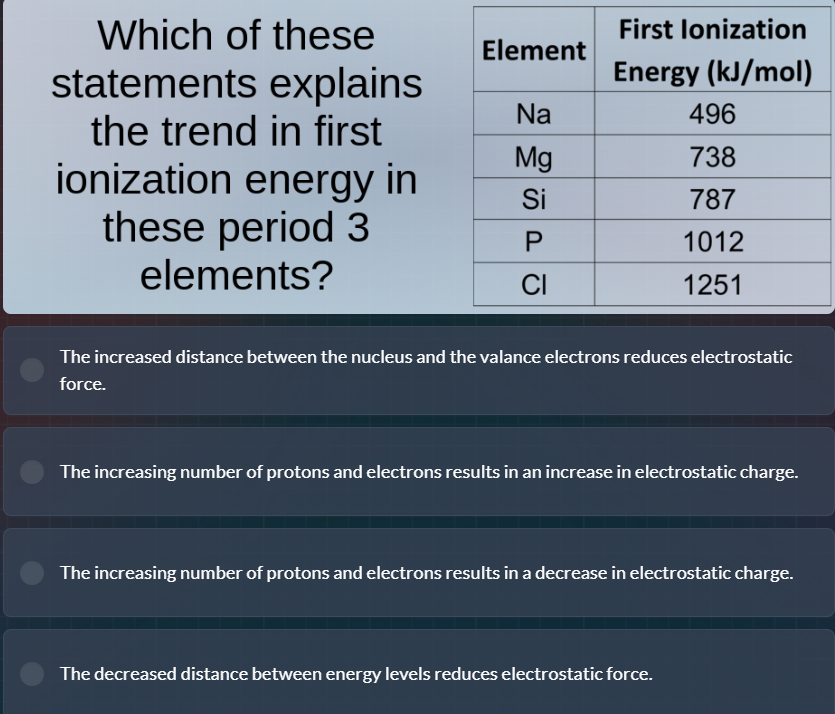
which of these statements explains the trend in first ionization energy in these period 3 elements?element first ionization energy (kj/mol)na 496mg 738si 787p 1012cl 1251the increased distance between the nucleus and the valance electrons reduces electrostatic force.the increasing number of protons and electrons results in an increase in electrostatic charge.the increasing number of protons and electrons results in a decrease in electrostatic charge.the decreased distance between energy levels reduces electrostatic force.
First ionization energy generally increases across a period because the number of protons (nuclear charge) increases while electrons are added to the same valence shell. This stronger positive charge from the nucleus creates a greater electrostatic attraction to the valence electrons, requiring more energy to remove an electron. The correct statement aligns with this trend, while the other options incorrectly describe electrostatic force/charge changes or distance effects that do not match period 3 behavior.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The increasing number of protons and electrons results in an increase in electrostatic charge.