QUESTION IMAGE
Question
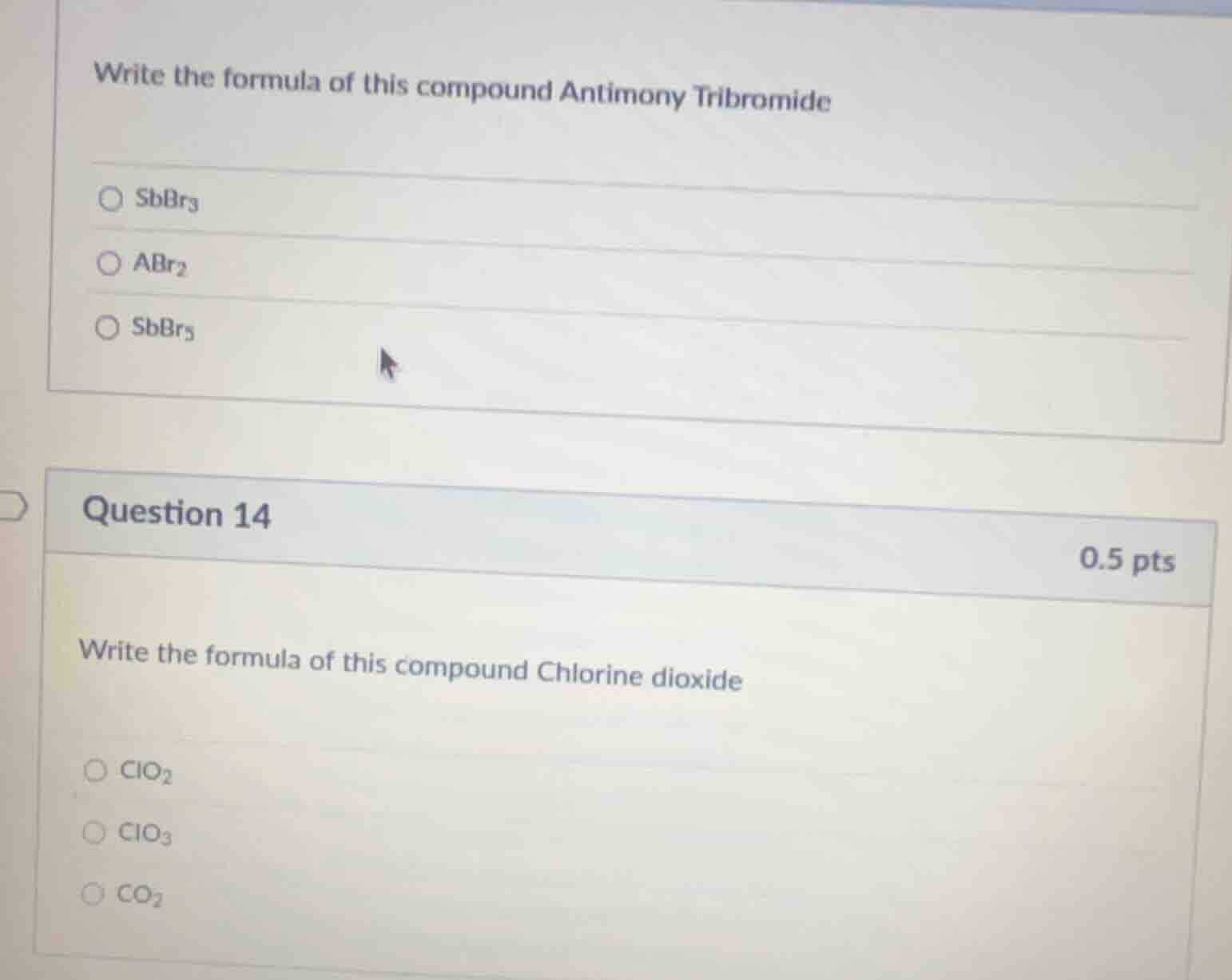
write the formula of this compound antimony tribromide
○ sbbr₃
○ abr₂
○ sbbr₅
question 14
0.5 pts
write the formula of this compound chlorine dioxide
○ clo₂
○ clo₃
○ co₂
Response
Antimony Tribromide Formula Question
Brief Explanations
Antimony (Sb) has a common oxidation state that pairs with three bromide (Br⁻) ions in "tribromide" (tri - 3). So the formula should be \( \text{SbBr}_3 \), matching the first option.
Brief Explanations
"Chlorine dioxide" means one chlorine (Cl) and two oxygen (O) atoms (di - 2 for oxygen). The formula is \( \text{ClO}_2 \), which is the first option.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \( \text{SbBr}_3 \)