QUESTION IMAGE
Question
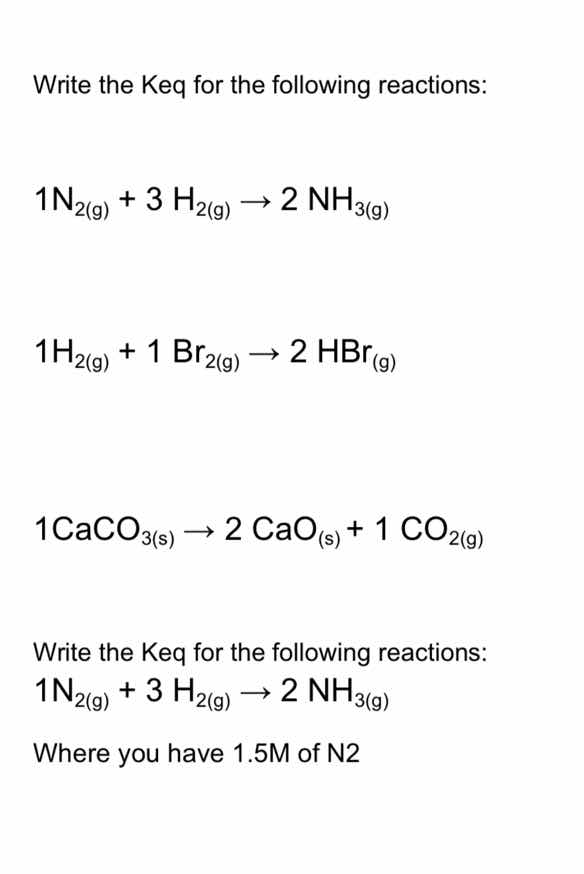
write the keq for the following reactions:
1n₂(g) + 3 h₂(g) → 2 nh₃(g)
1h₂(g) + 1 br₂(g) → 2 hbr(g)
1caco₃(s) → 2 cao(s) + 1 co₂(g)
write the keq for the following reactions:
1n₂(g) + 3 h₂(g) → 2 nh₃(g)
where you have 1.5m of n2
Step1: Define Keq for gas reactions
For a reaction $aA_{(g)} + bB_{(g)}
ightarrow cC_{(g)} + dD_{(g)}$, $K_{eq} = \frac{[products]}{[reactants]} = \frac{[C]^c[D]^d}{[A]^a[B]^b}$. Pure solids are excluded.
Step2: Keq for first reaction
Reaction: $\text{N}_{2(g)} + 3\text{H}_{2(g)}
ightarrow 2\text{NH}_{3(g)}$
$K_{eq} = \frac{[\text{NH}_3]^2}{[\text{N}_2][\text{H}_2]^3}$
Step3: Keq for second reaction
Reaction: $\text{H}_{2(g)} + \text{Br}_{2(g)}
ightarrow 2\text{HBr}_{(g)}$
$K_{eq} = \frac{[\text{HBr}]^2}{[\text{H}_2][\text{Br}_2]}$
Step4: Keq for third reaction
Reaction: $\text{CaCO}_{3(s)}
ightarrow \text{CaO}_{(s)} + \text{CO}_{2(g)}$
$K_{eq} = [\text{CO}_2]$
Step5: Keq for repeated first reaction
Given $[\text{N}_2]=1.5\text{M}$, substitute into the Keq expression:
$K_{eq} = \frac{[\text{NH}_3]^2}{1.5 \times [\text{H}_2]^3}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $K_{eq} = \frac{[\text{NH}_3]^2}{[\text{N}_2][\text{H}_2]^3}$
- $K_{eq} = \frac{[\text{HBr}]^2}{[\text{H}_2][\text{Br}_2]}$
- $K_{eq} = [\text{CO}_2]$
- $K_{eq} = \frac{[\text{NH}_3]^2}{1.5[\text{H}_2]^3}$