QUESTION IMAGE
Question
writing chemical formulas
question of the day? what number is never used as a subscript in a chemical formula?
science career? pharmacist
chemical formulas represent compounds.
oxidation numbers are used to determine the ratio in which elements combine to form compounds.
oxidation numbers tell the number of electrons an atom gained or lost when forming the compound.
- the plus or minus indicates if electrons were lost or gained.
- since electrons have a negative charge, and atom with a negative oxidation number will gain electrons. that means an atom with a positive oxidation number will lose electrons.
tip
nonmetals and polyatomic ions almost always gain electrons - have negative oxidation numbers.
metals almost always lose electrons - have positive oxidation numbers.
- the number indicates how many electrons.
example:
- aluminum has an oxidation number of +3. it will lose 3 electrons when forming compounds.
- oxygen has an oxidation number of -2. it will gain 2 electrons when forming compounds.
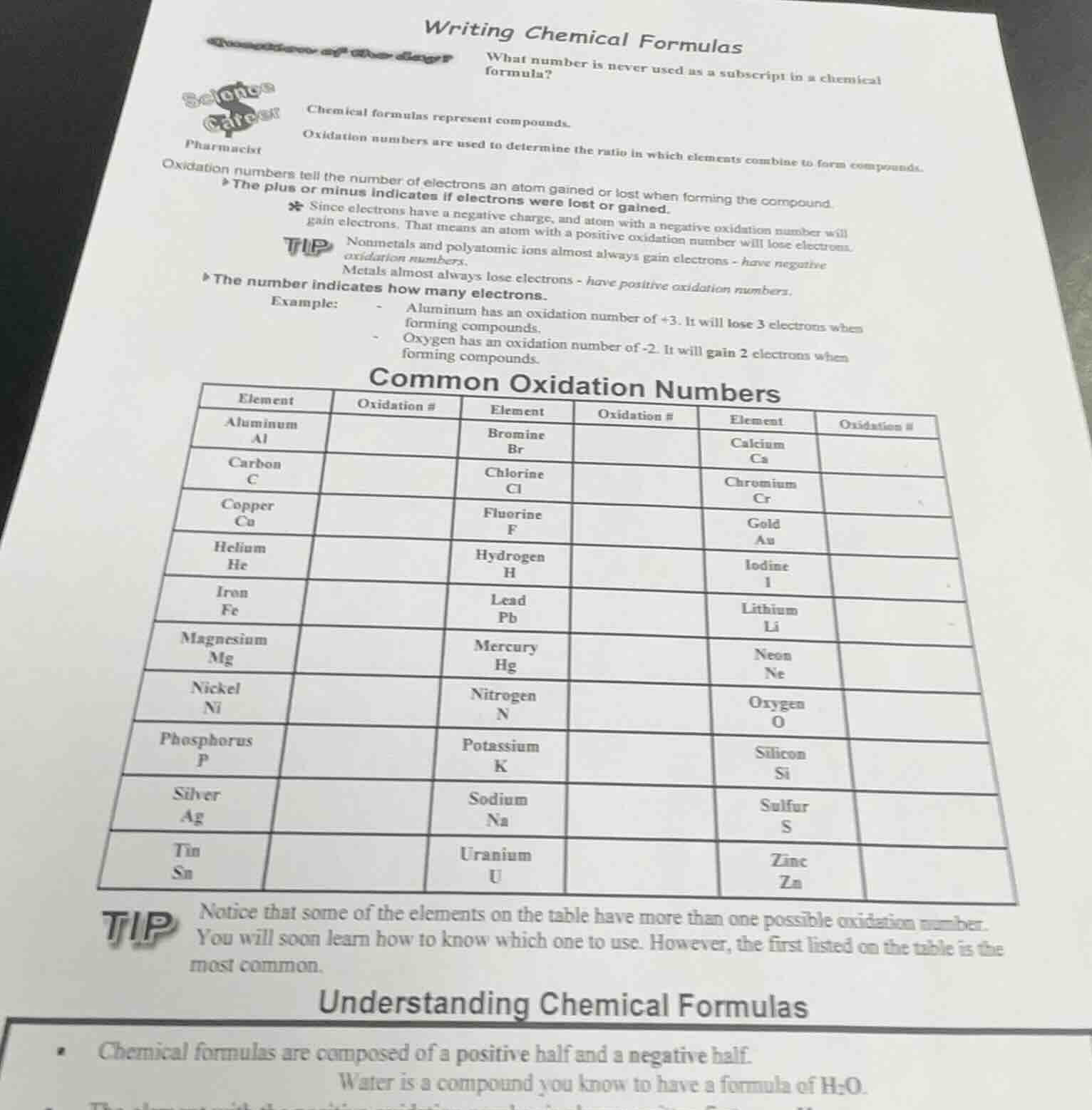
common oxidation numbers
chart with columns: element, oxidation #, element, oxidation #, element, oxidation #; rows list elements like aluminum (al), carbon (c), copper (cu), etc., with empty oxidation # cells
tip
notice that some of the elements on the table have more than one possible oxidation number. you will soon learn how to know which one to use. however, the first listed on the table is the most common.
understanding chemical formulas
- chemical formulas are composed of a positive half and a negative half.
water is a compound you know to have a formula of h₂o.
To determine the number never used as a subscript in a chemical formula, we analyze subscript rules. Subscripts represent the ratio of atoms in a compound, showing the number of each atom. A subscript of 1 is never written (e.g., H₂O has 2 H and 1 O, but O's subscript 1 is omitted). So the number is 1.
Step1: Recall subscript purpose
Subscripts show atom ratios in compounds.
Step2: Analyze subscript usage
Subscripts ≥1, but 1 is implied (not written). For example, in H₂O, O has subscript 1 (omitted), H has 2. So 1 is never used as a written subscript.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The number never used as a subscript in a chemical formula is 1.