QUESTION IMAGE
Question
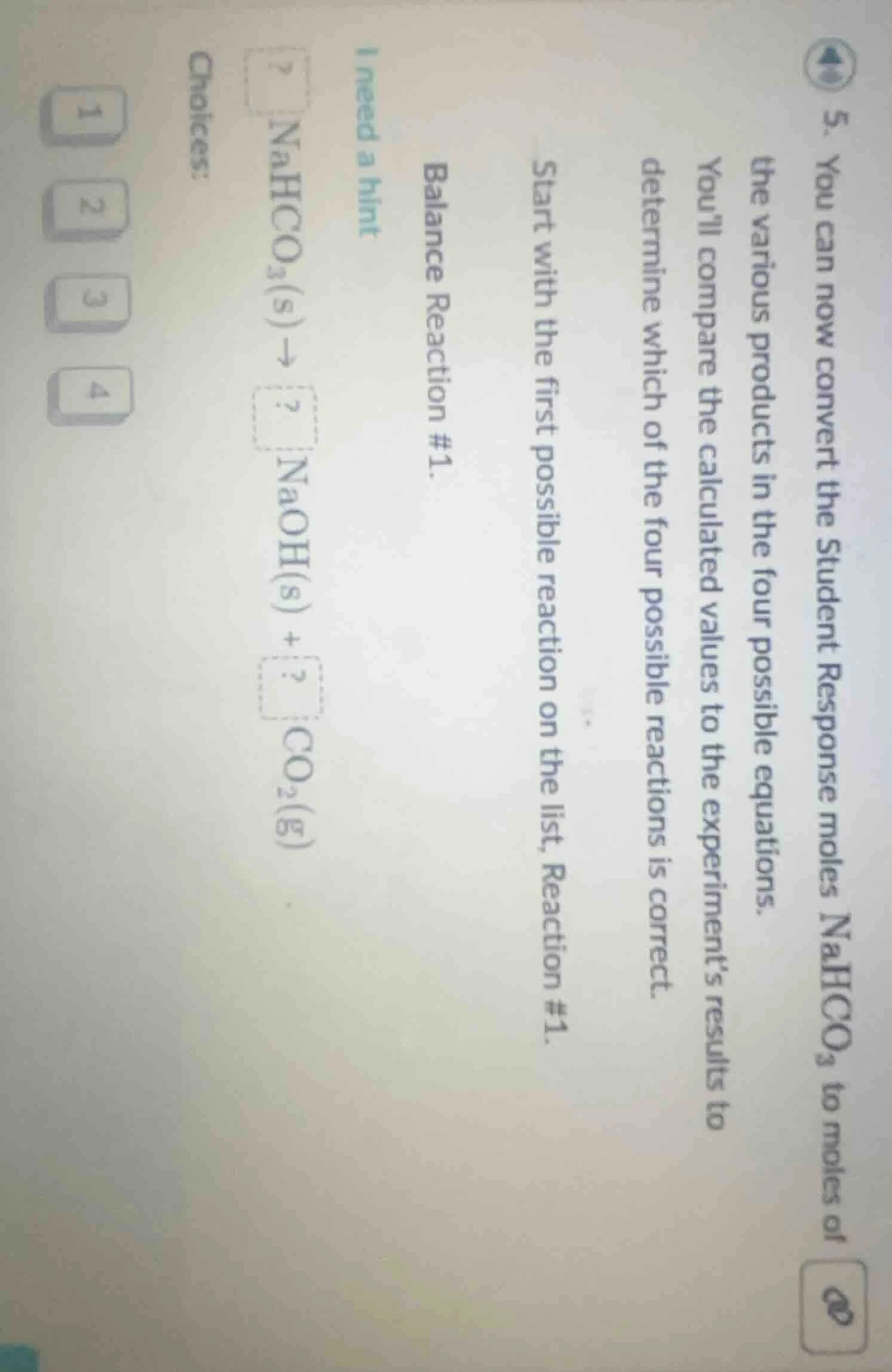
- you can now convert the student response moles $ce{nahco_{3}}$ to moles of the various products in the four possible equations.
youll compare the calculated values to the experiments results to determine which of the four possible reactions is correct.
start with the first possible reaction on the list, reaction #1.
balance reaction #1.
$\boldsymbol{?} ce{nahco_{3}(s)
ightarrow ? naoh(s) + ? co_{2}(g)}$
i need a hint
choices:
1 2 3 4
Step1: Count Na atoms
Left: 1 Na in $\text{NaHCO}_3$; Right: 1 Na in $\text{NaOH}$ (temporarily balanced)
Step2: Count C atoms
Left: 1 C in $\text{NaHCO}_3$; Right: 1 C in $\text{CO}_2$ (balanced)
Step3: Count H atoms
Left: 1 H in $\text{NaHCO}_3$; Right: 1 H in $\text{NaOH}$ (balanced)
Step4: Count O atoms
Left: 3 O in $\text{NaHCO}_3$; Right: 1 O in $\text{NaOH}$ + 2 O in $\text{CO}_2$ = 3 O (balanced)
Step5: Verify all atoms
All elements have equal counts on both sides.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\text{NaHCO}_3(s)
ightarrow \text{NaOH}(s) + \text{CO}_2(g)$
The coefficients are: 1, 1, 1