QUESTION IMAGE
Question
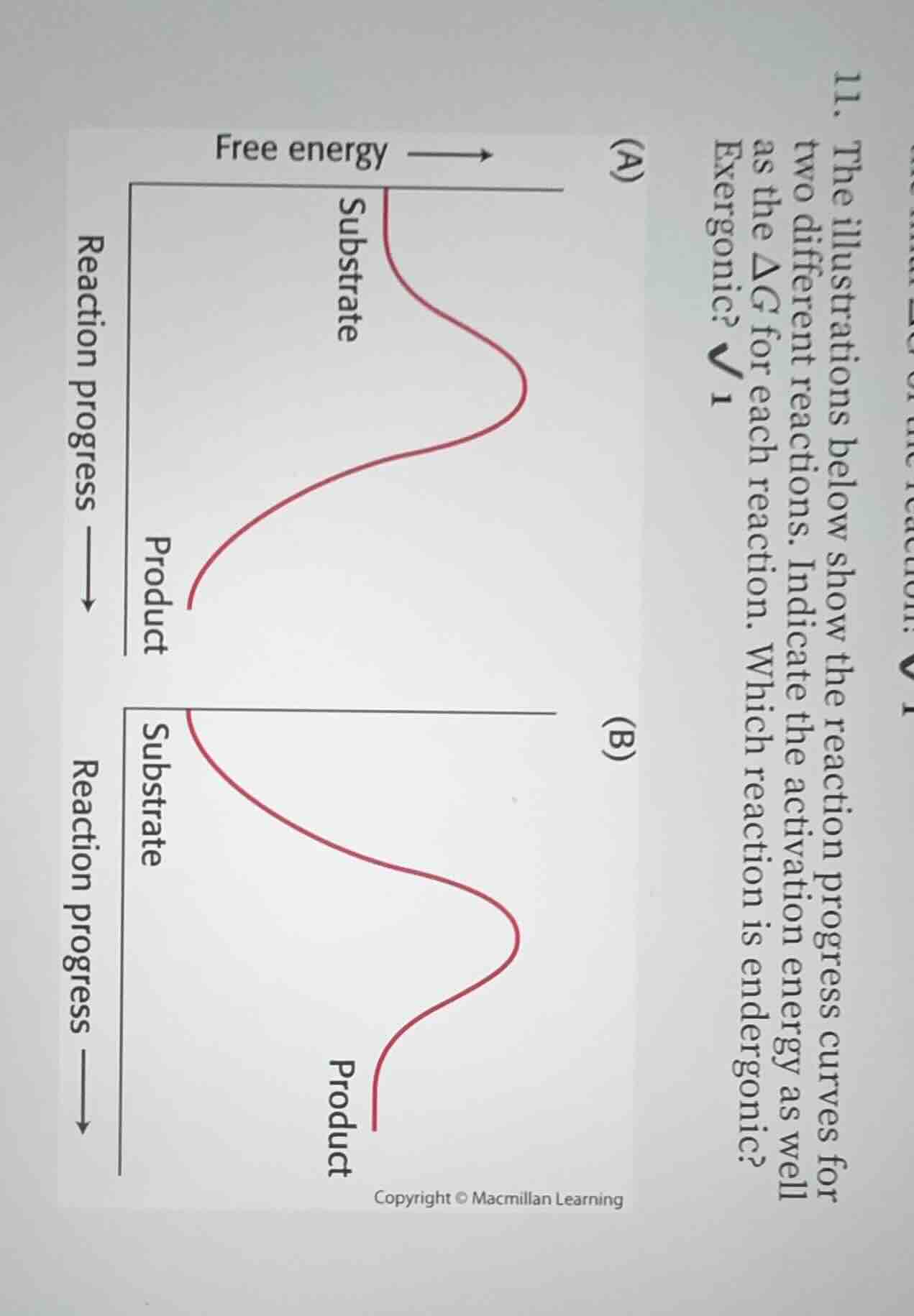
- the illustrations below show the reaction progress curves for two different reactions. indicate the activation energy as well as the $\delta g$ for each reaction. which reaction is endergonic? which reaction is exergonic? \\(\boldsymbol{\sqrt{}}_1\\)
Step1: Define activation energy
Activation energy ($E_a$) is the free energy difference between the substrate and the highest energy peak (transition state) on the curve.
For (A): $E_a = G_{\text{transition state (A)}} - G_{\text{substrate (A)}}$
For (B): $E_a = G_{\text{transition state (B)}} - G_{\text{substrate (B)}}$
Step2: Define $\Delta G$ (free energy change)
$\Delta G = G_{\text{product}} - G_{\text{substrate}}$
For (A): $\Delta G = G_{\text{product (A)}} - G_{\text{substrate (A)}} < 0$ (product has lower free energy)
For (B): $\Delta G = G_{\text{product (B)}} - G_{\text{substrate (B)}} > 0$ (product has higher free energy)
Step3: Classify reaction type
Exergonic reactions have $\Delta G < 0$; endergonic reactions have $\Delta G > 0$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Activation Energy:
- For Reaction (A): Activation energy is the vertical difference between the substrate free energy and the highest free energy peak on curve (A).
- For Reaction (B): Activation energy is the vertical difference between the substrate free energy and the highest free energy peak on curve (B).
- $\Delta G$:
- Reaction (A): $\Delta G = G_{\text{product}} - G_{\text{substrate}} < 0$ (negative value)
- Reaction (B): $\Delta G = G_{\text{product}} - G_{\text{substrate}} > 0$ (positive value)
- Reaction Classification:
- Reaction (A) is exergonic.
- Reaction (B) is endergonic.