QUESTION IMAGE
Question
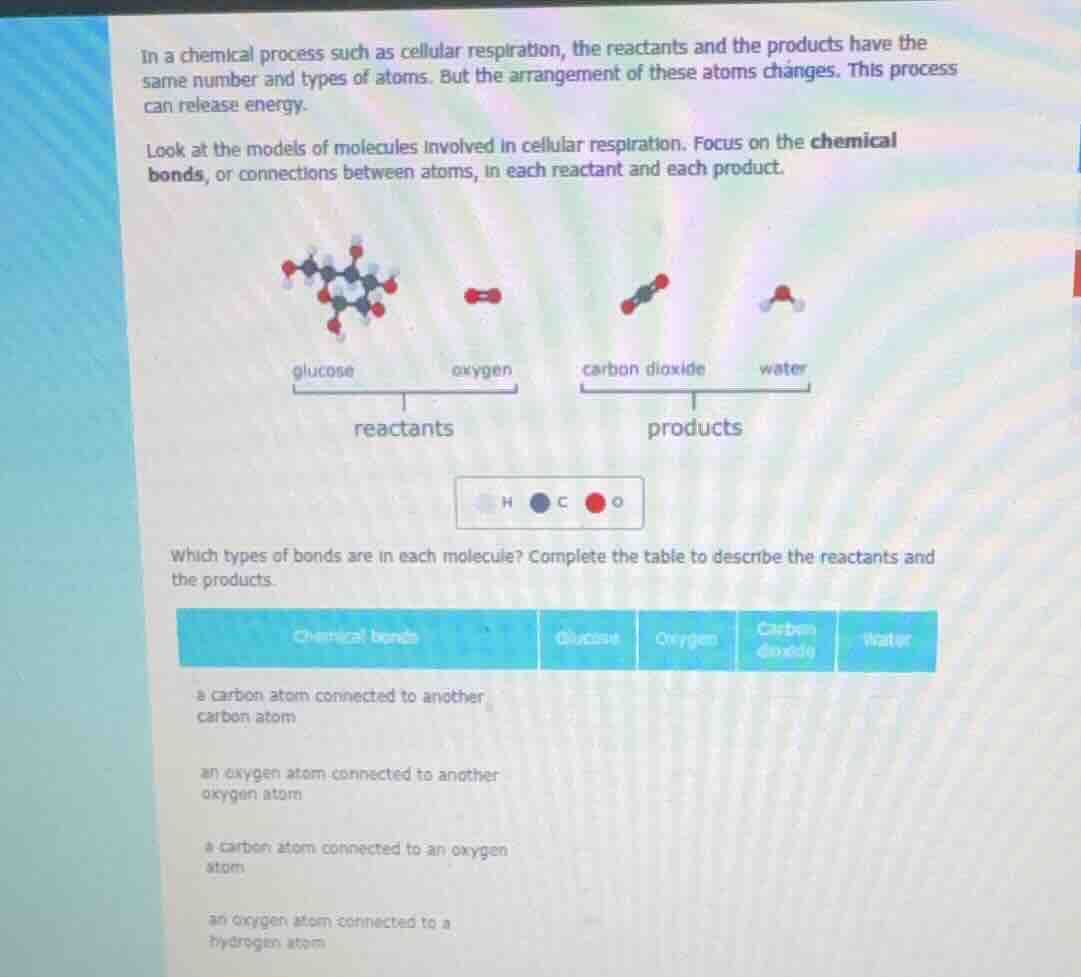
in a chemical process such as cellular respiration, the reactants and the products have the same number and types of atoms. but the arrangement of these atoms changes. this process can release energy. look at the models of molecules involved in cellular respiration. focus on the chemical bonds, or connections between atoms, in each reactant and each product. images of glucose, oxygen, carbon dioxide, water molecules with labels reactants and products, and a key for h, c, o atoms which types of bonds are in each molecule? complete the table to describe the reactants and the products. table with chemical bonds, glucose, oxygen, carbon dioxide, water columns and bond descriptions: a carbon atom connected to another carbon atom; an oxygen atom connected to another oxygen atom; a carbon atom connected to an oxygen atom; an oxygen atom connected to a hydrogen atom
- For glucose ($\text{C}_6\text{H}_{12}\text{O}_6$): Its structure has carbon-carbon chains, carbon-oxygen bonds (in hydroxyl/ketone groups), and oxygen-hydrogen bonds (in hydroxyl groups).
- For oxygen ($\text{O}_2$): It is a diatomic molecule with only oxygen-oxygen bonds.
- For carbon dioxide ($\text{CO}_2$): It has two carbon-oxygen double bonds, no other bond types here.
- For water ($\text{H}_2\text{O}$): It consists of oxygen-hydrogen single bonds, no other bond types here.
We mark "Yes" if the bond exists in the molecule, "No" if not.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Chemical bonds | Glucose | Oxygen | Carbon dioxide | Water |
|---|---|---|---|---|
| an oxygen atom connected to another oxygen atom | No | Yes | No | No |
| a carbon atom connected to an oxygen atom | Yes | No | Yes | No |
| an oxygen atom connected to a hydrogen atom | Yes | No | No | Yes |