QUESTION IMAGE
Question
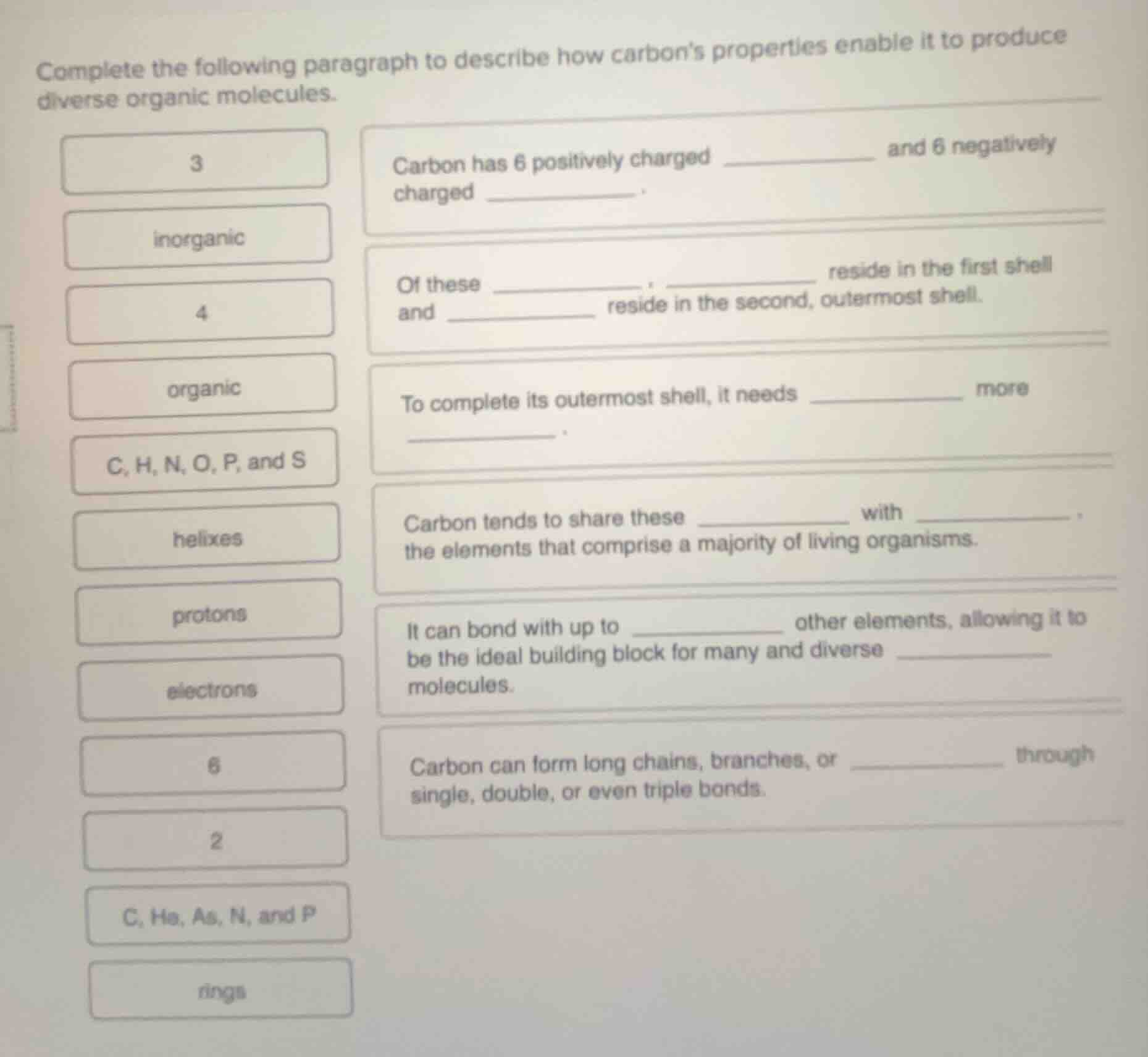
complete the following paragraph to describe how carbons properties enable it to produce diverse organic molecules.
carbon has 6 positively charged ________ and 6 negatively charged ________.
of these ________, ______ reside in the first shell and ________ reside in the second, outermost shell.
to complete its outermost shell, it needs ________ more ________.
carbon tends to share these ________ with ________ the elements that comprise a majority of living organisms.
it can bond with up to ________ other elements, allowing it to be the ideal building block for many and diverse ________ molecules.
carbon can form long chains, branches, or __________ through single, double, or even triple bonds.
options (from left column): 3, inorganic, 4, organic, c, h, n, o, p, and s, helixes, protons, electrons, 6, 2, c, he, as, n, and p, rings
- Carbon has 6 protons (positively charged particles in the nucleus) and 6 electrons (negatively charged particles orbiting the nucleus).
- Electrons are distributed in shells: the first shell holds 2 electrons, the second (outermost for carbon) holds 4.
- To fill its outermost shell (which can hold 8), carbon needs 4 more electrons.
- Carbon shares electrons with elements like C, H, N, O, P, S (major elements in living organisms).
- Carbon can bond with up to 4 other elements, making it ideal for organic molecules (since organic molecules are carbon - based).
- Carbon can form long chains, branches, or rings.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Carbon has 6 positively charged \(\boldsymbol{\text{protons}}\) and 6 negatively charged \(\boldsymbol{\text{electrons}}\).
- Of these \(\boldsymbol{\text{electrons}}\), \(\boldsymbol{2}\) reside in the first shell and \(\boldsymbol{4}\) reside in the second, outermost shell.
- To complete its outermost shell, it needs \(\boldsymbol{4}\) more \(\boldsymbol{\text{electrons}}\).
- Carbon tends to share these \(\boldsymbol{\text{electrons}}\) with \(\boldsymbol{\text{C, H, N, O, P, and S}}\).
- It can bond with up to \(\boldsymbol{4}\) other elements, allowing it to be the ideal building block for many and diverse \(\boldsymbol{\text{organic}}\) molecules.
- Carbon can form long chains, branches, or \(\boldsymbol{\text{rings}}\) through single, double, or even triple bonds.