QUESTION IMAGE
Question
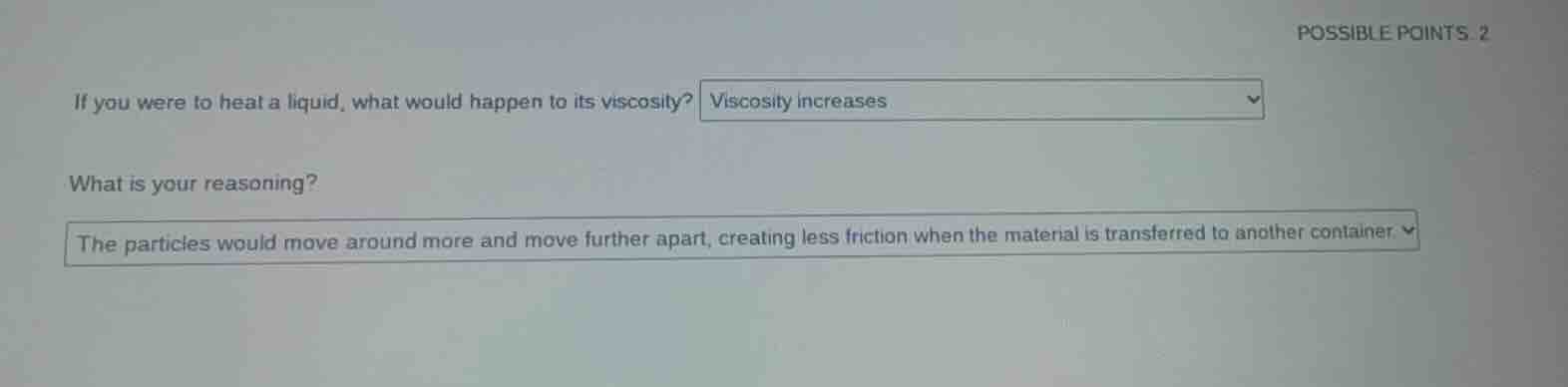
possible points. 2
if you were to heat a liquid, what would happen to its viscosity? viscosity increases
what is your reasoning?
the particles would move around more and move further apart, creating less friction when the material is transferred to another container.
The initial selected answer is incorrect. For liquids, heating increases the kinetic energy of their particles, which weakens the intermolecular forces that hold the particles together. This allows the particles to move past one another more easily, reducing the internal friction (viscosity) of the liquid. The provided reasoning incorrectly states particles move further apart (a more significant change in gases) and mislinks to friction during container transfer; the core reason is reduced intermolecular cohesion in liquids when heated.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Viscosity decreases
- Heating increases liquid particle kinetic energy, weakening intermolecular forces, reducing internal friction/viscosity.