QUESTION IMAGE
Question
question 11 (1 point)
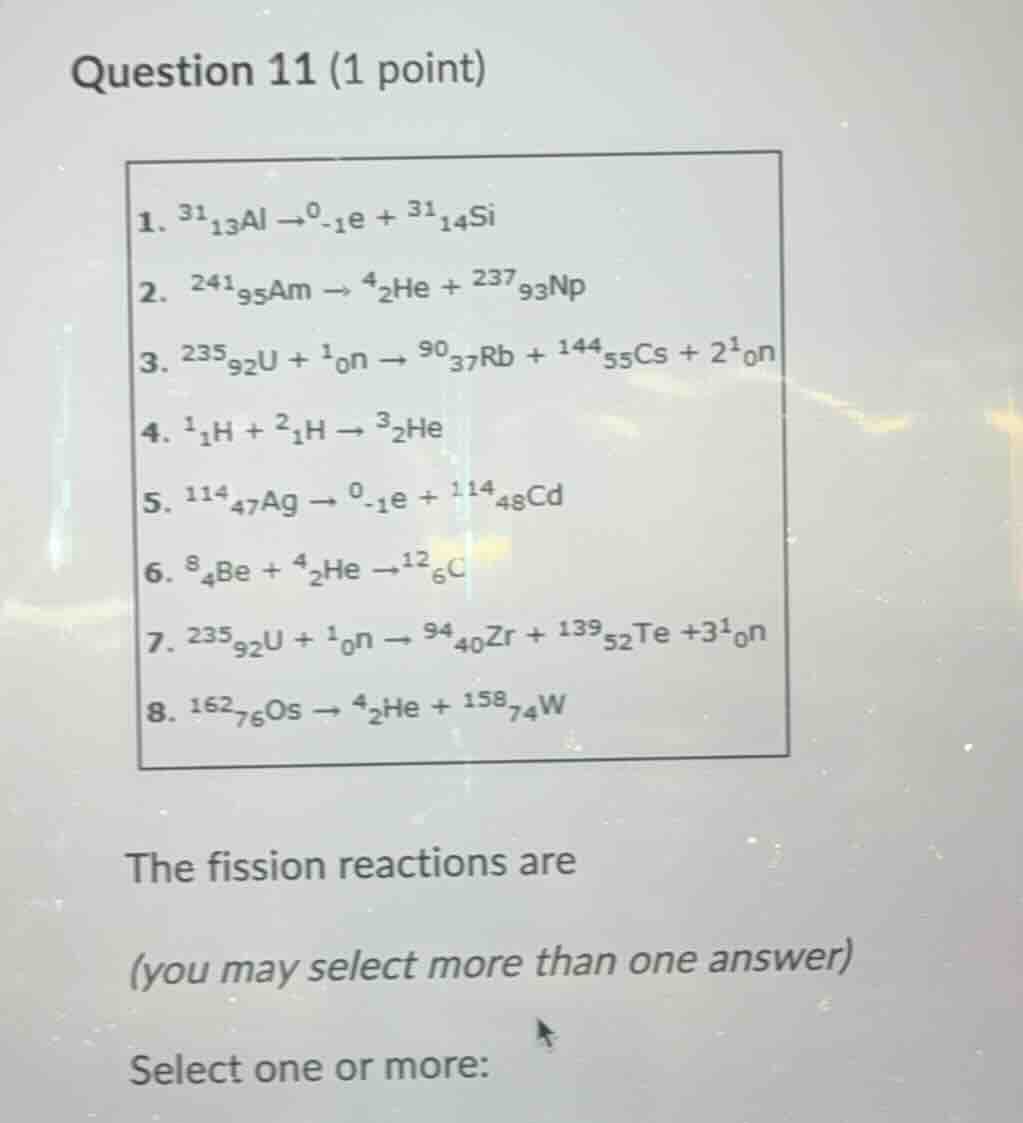
- $^{31}_{13}\text{al} \
ightarrow ^{0}_{-1}\text{e} + ^{31}_{14}\text{si}$
- $^{241}_{95}\text{am} \
ightarrow ^{4}_{2}\text{he} + ^{237}_{93}\text{np}$
- $^{235}_{92}\text{u} + ^{1}_{0}\text{n} \
ightarrow ^{90}_{37}\text{rb} + ^{144}_{55}\text{cs} + 2^{1}_{0}\text{n}$
- $^{1}_{1}\text{h} + ^{2}_{1}\text{h} \
ightarrow ^{3}_{2}\text{he}$
- $^{114}_{47}\text{ag} \
ightarrow ^{0}_{-1}\text{e} + ^{114}_{48}\text{cd}$
- $^{8}_{4}\text{be} + ^{4}_{2}\text{he} \
ightarrow ^{12}_{6}\text{c}$
- $^{235}_{92}\text{u} + ^{1}_{0}\text{n} \
ightarrow ^{94}_{40}\text{zr} + ^{139}_{52}\text{te} + 3^{1}_{0}\text{n}$
- $^{162}_{76}\text{os} \
ightarrow ^{4}_{2}\text{he} + ^{158}_{74}\text{w}$
the fission reactions are
(you may select more than one answer)
select one or more:
Step1: Define nuclear fission
Nuclear fission is a reaction where a heavy nucleus splits into smaller lighter nuclei, often with neutron release, initiated by a neutron bombardment.
Step2: Analyze each reaction
- Beta decay (nucleus emits electron, mass stays same)
- Alpha decay (nucleus emits $^4_2\text{He}$)
- Heavy U-235 splits into Rb-90 + Cs-144, neutrons released: fits fission.
- Fusion (light nuclei combine into heavier one)
- Beta decay (nucleus emits electron)
- Fusion (Be + He combine into C)
- Heavy U-235 splits into Zr-94 + Te-139, neutrons released: fits fission.
- Alpha decay (nucleus emits $^4_2\text{He}$)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $^{235}_{92}\text{U} + ^{1}_{0}\text{n}
ightarrow ^{90}_{37}\text{Rb} + ^{144}_{55}\text{Cs} + 2^{1}_{0}\text{n}$, 7. $^{235}_{92}\text{U} + ^{1}_{0}\text{n}
ightarrow ^{94}_{40}\text{Zr} + ^{139}_{52}\text{Te} +3^{1}_{0}\text{n}$