QUESTION IMAGE
Question
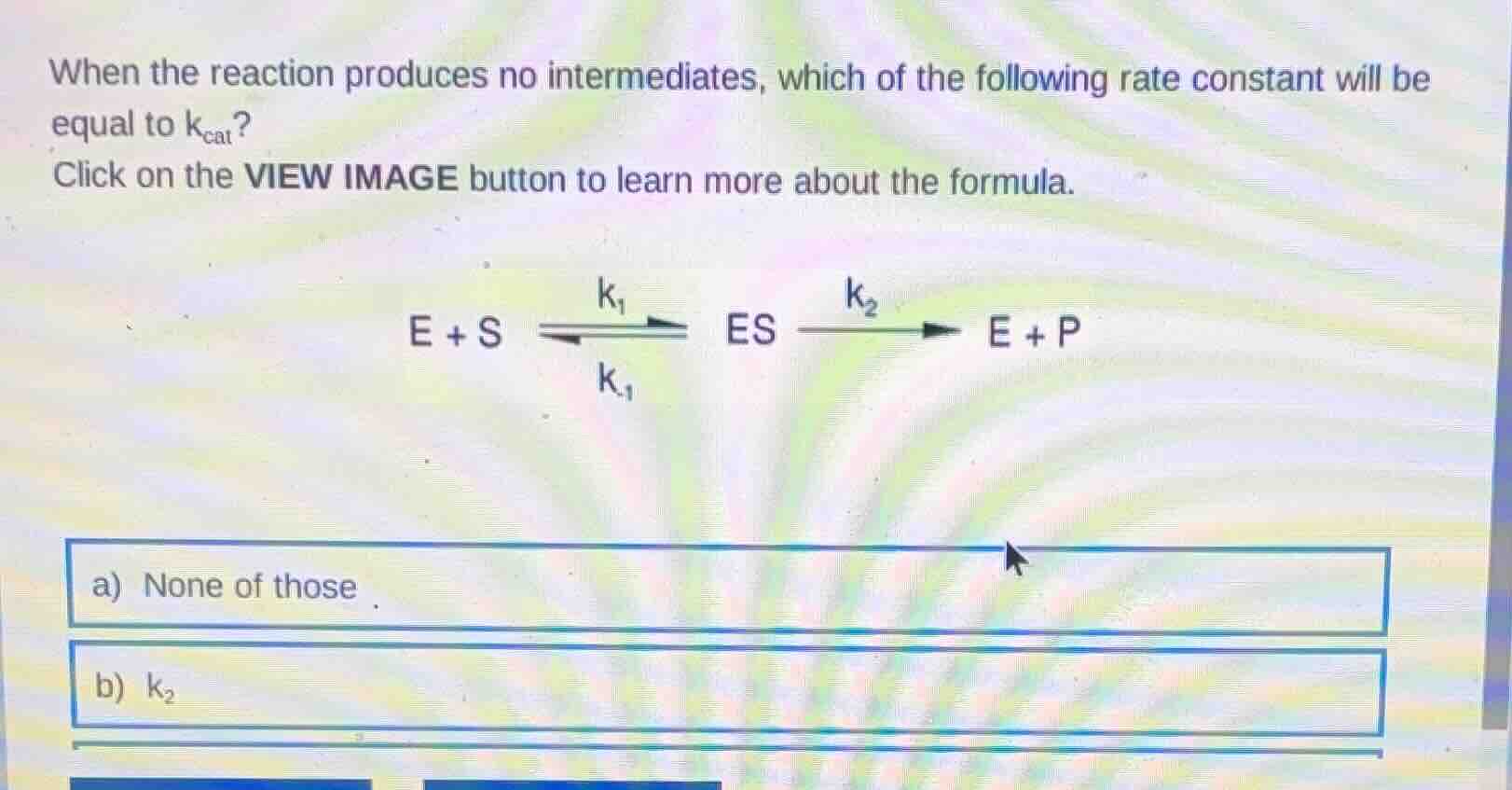
when the reaction produces no intermediates, which of the following rate constant will be equal to $k_{cat}$?
click on the view image button to learn more about the formula.
$e + s \xrightleftharpoonsk_{-1}{k_1} es \xrightarrow{k_2} e + p$
a) none of those
b) $k_2$
$k_{cat}$ (turnover number) represents the rate of the catalytic step where the enzyme-substrate complex (ES) converts to product (P) and free enzyme (E). In the given reaction scheme, this step is governed by the rate constant $k_2$, and there are no additional intermediates in the catalytic pathway, so $k_{cat}$ equals $k_2$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
b) $k_2$