QUESTION IMAGE
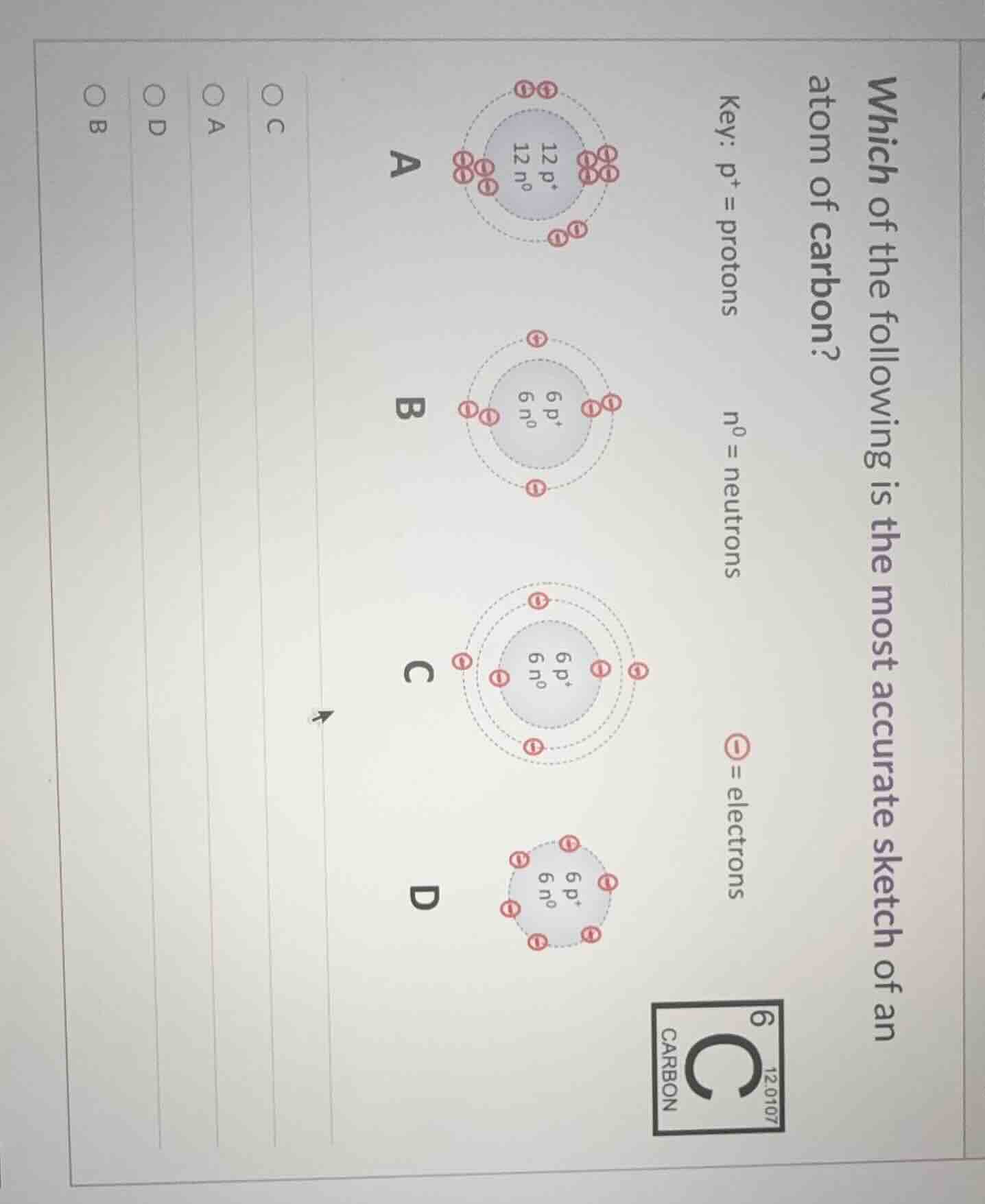
Question
which of the following is the most accurate sketch of an atom of carbon?
key: p⁺ = protons
n⁰ = neutrons
⊖ = electrons
a: 12 p⁺, 12 n⁰
b: 6 p⁺, 6 n⁰
c: 6 p⁺, 6 n⁰
d: 6 p⁺, 6 n⁰
○ c
○ a
○ d
○ b
Step1: Confirm carbon's proton count
From the periodic table, carbon has an atomic number of 6, so it has $6\ p^+$. This eliminates option A (12 protons).
Step2: Confirm neutral atom electron count
A neutral carbon atom has equal protons and electrons, so $6\ e^-$. This eliminates option B (5 electrons) and D (5 electrons).
Step3: Confirm neutron count
Carbon's mass number is ~12, so neutrons = $12 - 6 = 6\ n^0$, which matches option C.
Step4: Verify electron shell arrangement
Carbon has 2 electrons in the first shell and 4 in the second, which matches option C's structure.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C