QUESTION IMAGE
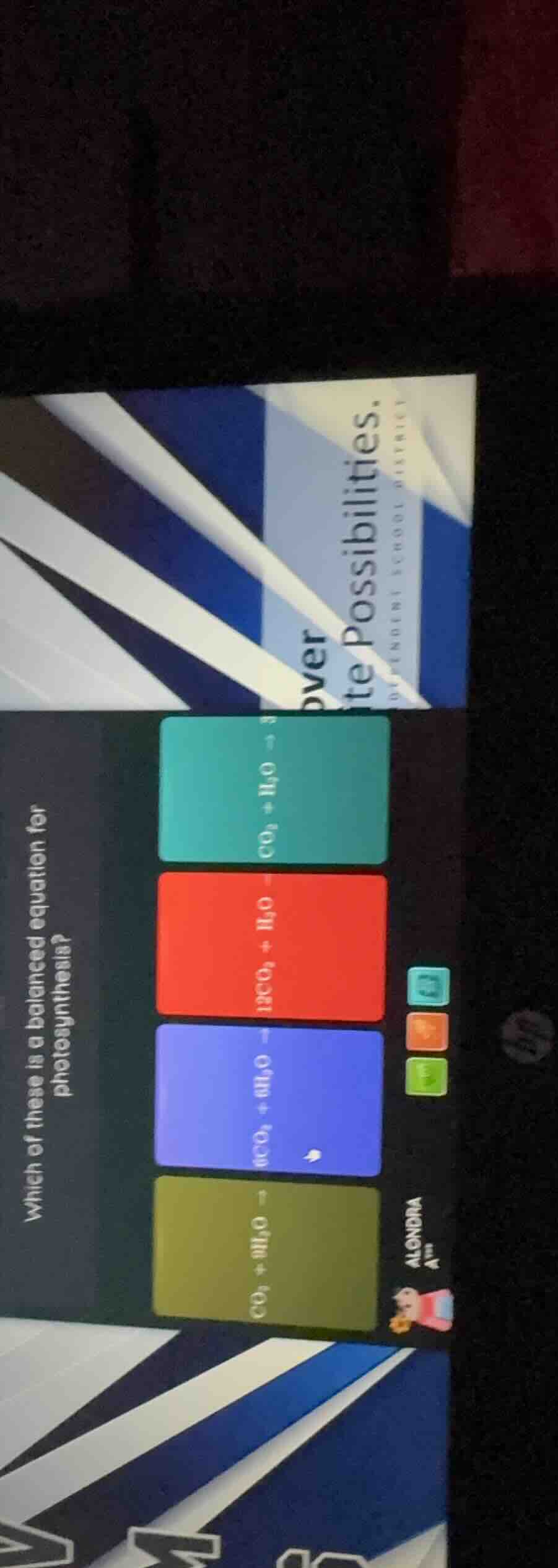
Question
which of these is a balanced equation for photosynthesis? \
options with chemical equations, some text like over tepossibilities and glendale school district and a user alondra with an avatar
To determine the balanced equation for photosynthesis, we first recall the correct balanced chemical equation for photosynthesis: \( 6CO_2 + 6H_2O \xrightarrow[\text{Chlorophyll}]{\text{Sunlight}} C_6H_{12}O_6 + 6O_2 \). Wait, but looking at the options (even with some text clarity issues, likely the options are about balancing \( CO_2 \), \( H_2O \), and products). Wait, maybe the options are like checking the number of \( C \), \( H \), \( O \) atoms. Let's assume the correct balanced one (maybe the blue or other, but actually the standard photosynthesis equation has 6 \( CO_2 \), 6 \( H_2O \) yielding \( C_6H_{12}O_6 \) and 6 \( O_2 \). But if the options are simplified (maybe showing reactants and products with correct coefficients), let's check:
Wait, the user's image has options, maybe the correct one is the one with 6 \( CO_2 \) and 6 \( H_2O \) (but the text is a bit unclear). Wait, maybe the red option? No, wait, the standard balanced equation for photosynthesis is \( 6CO_2 + 6H_2O
ightarrow C_6H_{12}O_6 + 6O_2 \). But if the options are like \( 6CO_2 + 6H_2O
ightarrow \) (with products), but maybe the blue option? Wait, maybe the correct answer is the one with 6 \( CO_2 \) and 6 \( H_2O \) (assuming the blue option has \( 6CO_2 + 6H_2O \) or similar). Wait, maybe the user's options: let's re-express.
Wait, the question is "Which of these is a balanced equation for photosynthesis?" The correct balanced chemical equation for photosynthesis is:
\( 6CO_2 + 6H_2O \xrightarrow{\text{Light Energy}} C_6H_{12}O_6 + 6O_2 \)
So we need to check which option has equal numbers of C, H, O on both sides. Let's assume the options:
- Teal: \( CO_2 + H_2O
ightarrow \) (unbalanced, 1 C, 2 O, 2 H on left; products would be unbalanced)
- Red: \( 12CO_2 + H_2O
ightarrow \) (no, too many C)
- Blue: \( 6CO_2 + 6H_2O
ightarrow \) (matches the balanced equation's reactants, leading to \( C_6H_{12}O_6 + 6O_2 \))
- Green: \( CO_2 + 6H_2O
ightarrow \) (unbalanced C)
So the correct option is likely the blue one (assuming the blue option has \( 6CO_2 + 6H_2O \) or the balanced coefficients).
But to confirm, the balanced equation for photosynthesis has 6 carbon dioxide molecules and 6 water molecules as reactants, producing 1 glucose and 6 oxygen molecules. So the option with 6 \( CO_2 \) and 6 \( H_2O \) (or the correct coefficients) is the balanced one.
The balanced chemical equation for photosynthesis is \( 6CO_2 + 6H_2O \xrightarrow{\text{Light Energy}} C_6H_{12}O_6 + 6O_2 \). We check the number of carbon (C), hydrogen (H), and oxygen (O) atoms on both sides. For the correct option, the number of C, H, O atoms in reactants ( \( CO_2 \) and \( H_2O \)) must equal those in products (glucose \( C_6H_{12}O_6 \) and \( O_2 \)). The option with 6 \( CO_2 \) and 6 \( H_2O \) (matching the balanced coefficients) is the correct balanced equation.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Assuming the blue option (or the one with \( 6CO_2 + 6H_2O \)) is the correct balanced equation for photosynthesis. (Note: Based on standard photosynthesis chemistry, the balanced equation has 6 \( CO_2 \), 6 \( H_2O \), 1 \( C_6H_{12}O_6 \), and 6 \( O_2 \).)