QUESTION IMAGE
Question
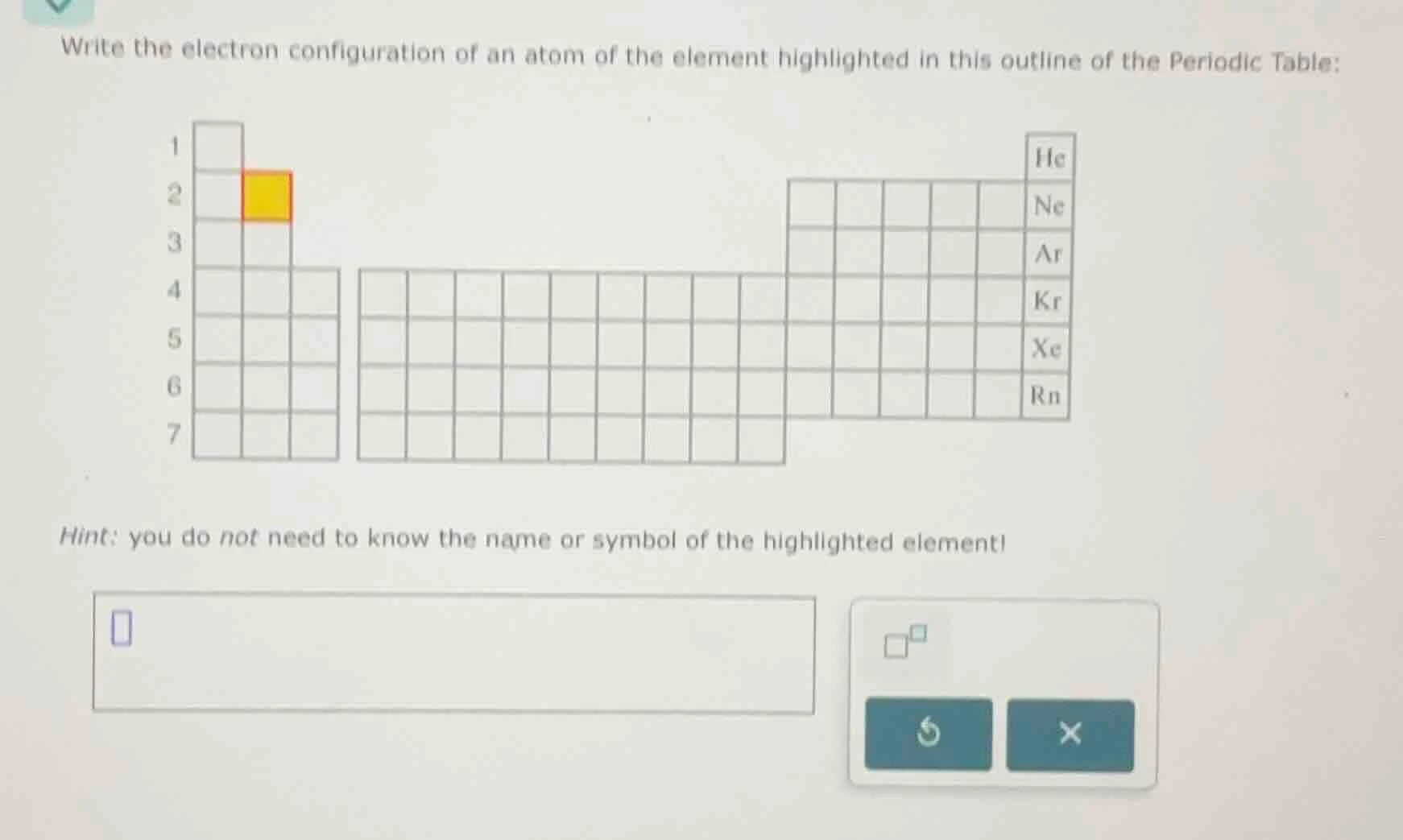
write the electron configuration of an atom of the element highlighted in this outline of the periodic table:
hint: you do not need to know the name or symbol of the highlighted element!
Step1: Identify element position
The highlighted element is in period 2, group 2 (2nd column, row 2), so it has an atomic number of 4.
Step2: Fill orbitals by Aufbau principle
First fill 1s orbital (max 2 electrons), then 2s orbital (remaining 2 electrons).
<Expression>
$1s^2 2s^2$
</Expression>
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$1s^2 2s^2$