QUESTION IMAGE
Question
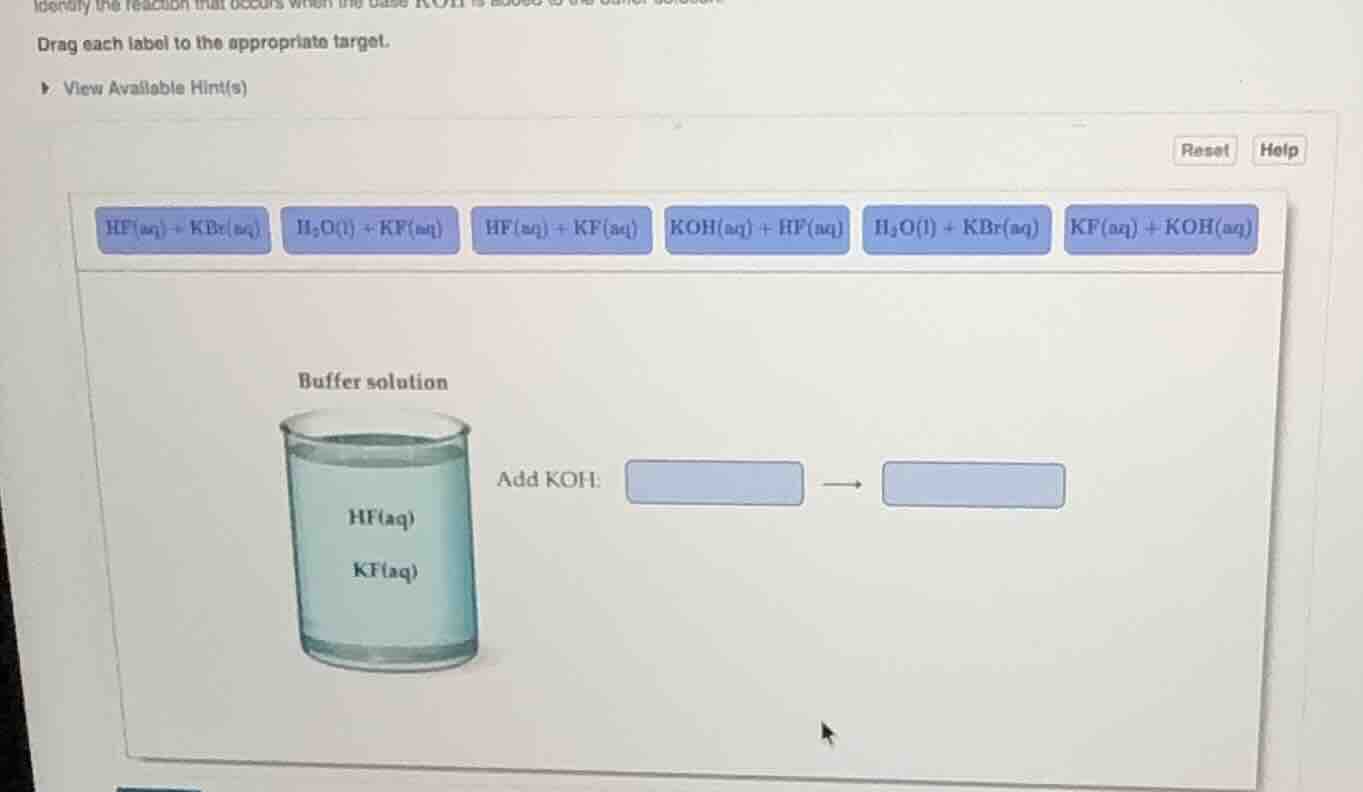
identify the reaction that occurs when the base koh is added to the buffer solution. drag each label to the appropriate target. view available hint(s) reset help hf(aq) + kbr(aq) h₂o(l) + kf(aq) hf(aq) + kf(aq) koh(aq) + hf(aq) h₂o(l) + kbr(aq) kf(aq) + koh(aq) buffer solution hf(aq) kf(aq) add koh:
Step1: Identify the reactants
The buffer solution contains \( \text{HF}(aq) \) and \( \text{KF}(aq) \), and we are adding \( \text{KOH}(aq) \). The base \( \text{KOH} \) will react with the acid \( \text{HF} \) in the buffer. So the reaction between the added \( \text{KOH} \) and the acid in the buffer is \( \text{KOH}(aq) + \text{HF}(aq) \).
Step2: Determine the products
When a strong base (\( \text{KOH} \)) reacts with a weak acid (\( \text{HF} \)), the products are water and the salt formed from the cation of the base and the anion of the acid. The reaction is a neutralization reaction: \( \text{KOH}(aq) + \text{HF}(aq)
ightarrow \text{H}_2\text{O}(l) + \text{KF}(aq) \). So the reactant side (before reaction) is \( \text{KOH}(aq) + \text{HF}(aq) \) and the product side (after reaction) is \( \text{H}_2\text{O}(l) + \text{KF}(aq) \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First box (reactant): \( \text{KOH}(aq) + \text{HF}(aq) \)
Second box (product): \( \text{H}_2\text{O}(l) + \text{KF}(aq) \)