QUESTION IMAGE
Question
name
class
date
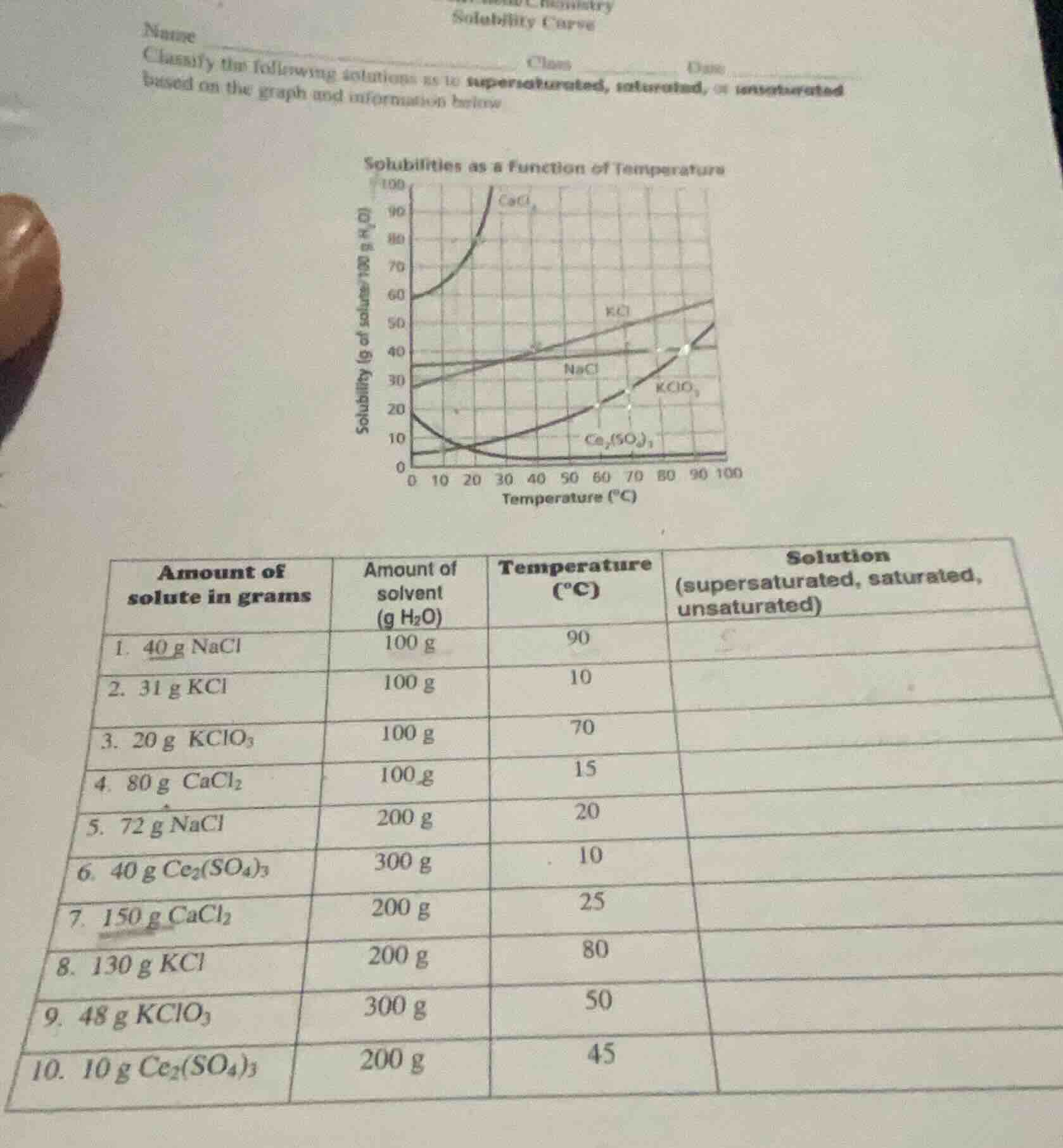
solubility curve
classify the following solutions as to supersaturated, saturated, or unsaturated
based on the graph and information below
solubilities as a function of temperature
amount of
solute in grams\tamount of
solvent
(g h₂o)\ttemperature
(°c)\tsolution
(supersaturated, saturated,
unsaturated)
- 40 g nacl\t100 g\t90\t
- 31 g kcl\t100 g\t10\t
- 20 g kclo₃\t100 g\t70\t
- 80 g cacl₂\t100 g\t15\t
- 72 g nacl\t200 g\t20\t
- 40 g ce₂(so₄)₃\t300 g\t10\t
- 150 g cacl₂\t200 g\t25\t
- 130 g kcl\t200 g\t80\t
- 48 g kclo₃\t300 g\t50\t
- 10 g ce₂(so₄)₃\t200 g\t45\t
To solve this, we analyze each solution by comparing the amount of solute to the solubility (from the curve) at the given temperature. A solution is:
- Unsaturated: Solute < Solubility
- Saturated: Solute = Solubility
- Supersaturated: Solute > Solubility (rare, often from cooling a saturated solution)
1. 40 g NaCl, 100 g H₂O, 90°C
- From the curve, NaCl solubility at 90°C ≈ 40 g/100 g H₂O.
- 40 g (solute) = 40 g (solubility).
- Classification: Saturated
2. 31 g KCl, 100 g H₂O, 10°C
- KCl solubility at 10°C ≈ 30 g/100 g H₂O.
- 31 g (solute) > 30 g (solubility).
- Classification: Supersaturated
3. 20 g KClO₃, 100 g H₂O, 70°C
- KClO₃ solubility at 70°C ≈ 30 g/100 g H₂O.
- 20 g (solute) < 30 g (solubility).
- Classification: Unsaturated
4. 80 g CaCl₂, 100 g H₂O, 15°C
- CaCl₂ solubility at 15°C ≈ 80 g/100 g H₂O (curve shows ~80 g at 15°C).
- 80 g (solute) = 80 g (solubility).
- Classification: Saturated
5. 72 g NaCl, 200 g H₂O, 20°C
- NaCl solubility at 20°C ≈ 36 g/100 g H₂O. For 200 g H₂O, solubility = \( 36 \times 2 = 72 \) g.
- 72 g (solute) = 72 g (solubility).
- Classification: Saturated
6. 40 g Ce₂(SO₄)₃, 300 g H₂O, 10°C
- Ce₂(SO₄)₃ solubility at 10°C ≈ 5 g/100 g H₂O. For 300 g H₂O, solubility = \( 5 \times 3 = 15 \) g.
- 40 g (solute) > 15 g (solubility).
- Classification: Supersaturated
7. 150 g CaCl₂, 200 g H₂O, 25°C
- CaCl₂ solubility at 25°C ≈ 80 g/100 g H₂O. For 200 g H₂O, solubility = \( 80 \times 2 = 160 \) g.
- 150 g (solute) < 160 g (solubility).
- Classification: Unsaturated
8. 130 g KCl, 200 g H₂O, 80°C
- KCl solubility at 80°C ≈ 50 g/100 g H₂O. For 200 g H₂O, solubility = \( 50 \times 2 = 100 \) g.
- 130 g (solute) > 100 g (solubility).
- Classification: Supersaturated
9. 48 g KClO₃, 300 g H₂O, 50°C
- KClO₃ solubility at 50°C ≈ 20 g/100 g H₂O. For 300 g H₂O, solubility = \( 20 \times 3 = 60 \) g.
- 48 g (solute) < 60 g (solubility).
- Classification: Unsaturated
10. 10 g Ce₂(SO₄)₃, 200 g H₂O, 45°C
- Ce₂(SO₄)₃ solubility at 45°C ≈ 5 g/100 g H₂O. For 200 g H₂O, solubility = \( 5 \times 2 = 10 \) g.
- 10 g (solute) = 10 g (solubility).
- Classification: Saturated
Final Answers:
- Saturated
- Supersaturated
- Unsaturated
- Saturated
- Saturated
- Supersaturated
- Unsaturated
- Supersaturated
- Unsaturated
- Saturated
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this, we analyze each solution by comparing the amount of solute to the solubility (from the curve) at the given temperature. A solution is:
- Unsaturated: Solute < Solubility
- Saturated: Solute = Solubility
- Supersaturated: Solute > Solubility (rare, often from cooling a saturated solution)
1. 40 g NaCl, 100 g H₂O, 90°C
- From the curve, NaCl solubility at 90°C ≈ 40 g/100 g H₂O.
- 40 g (solute) = 40 g (solubility).
- Classification: Saturated
2. 31 g KCl, 100 g H₂O, 10°C
- KCl solubility at 10°C ≈ 30 g/100 g H₂O.
- 31 g (solute) > 30 g (solubility).
- Classification: Supersaturated
3. 20 g KClO₃, 100 g H₂O, 70°C
- KClO₃ solubility at 70°C ≈ 30 g/100 g H₂O.
- 20 g (solute) < 30 g (solubility).
- Classification: Unsaturated
4. 80 g CaCl₂, 100 g H₂O, 15°C
- CaCl₂ solubility at 15°C ≈ 80 g/100 g H₂O (curve shows ~80 g at 15°C).
- 80 g (solute) = 80 g (solubility).
- Classification: Saturated
5. 72 g NaCl, 200 g H₂O, 20°C
- NaCl solubility at 20°C ≈ 36 g/100 g H₂O. For 200 g H₂O, solubility = \( 36 \times 2 = 72 \) g.
- 72 g (solute) = 72 g (solubility).
- Classification: Saturated
6. 40 g Ce₂(SO₄)₃, 300 g H₂O, 10°C
- Ce₂(SO₄)₃ solubility at 10°C ≈ 5 g/100 g H₂O. For 300 g H₂O, solubility = \( 5 \times 3 = 15 \) g.
- 40 g (solute) > 15 g (solubility).
- Classification: Supersaturated
7. 150 g CaCl₂, 200 g H₂O, 25°C
- CaCl₂ solubility at 25°C ≈ 80 g/100 g H₂O. For 200 g H₂O, solubility = \( 80 \times 2 = 160 \) g.
- 150 g (solute) < 160 g (solubility).
- Classification: Unsaturated
8. 130 g KCl, 200 g H₂O, 80°C
- KCl solubility at 80°C ≈ 50 g/100 g H₂O. For 200 g H₂O, solubility = \( 50 \times 2 = 100 \) g.
- 130 g (solute) > 100 g (solubility).
- Classification: Supersaturated
9. 48 g KClO₃, 300 g H₂O, 50°C
- KClO₃ solubility at 50°C ≈ 20 g/100 g H₂O. For 300 g H₂O, solubility = \( 20 \times 3 = 60 \) g.
- 48 g (solute) < 60 g (solubility).
- Classification: Unsaturated
10. 10 g Ce₂(SO₄)₃, 200 g H₂O, 45°C
- Ce₂(SO₄)₃ solubility at 45°C ≈ 5 g/100 g H₂O. For 200 g H₂O, solubility = \( 5 \times 2 = 10 \) g.
- 10 g (solute) = 10 g (solubility).
- Classification: Saturated
Final Answers:
- Saturated
- Supersaturated
- Unsaturated
- Saturated
- Saturated
- Supersaturated
- Unsaturated
- Supersaturated
- Unsaturated
- Saturated