QUESTION IMAGE
Question
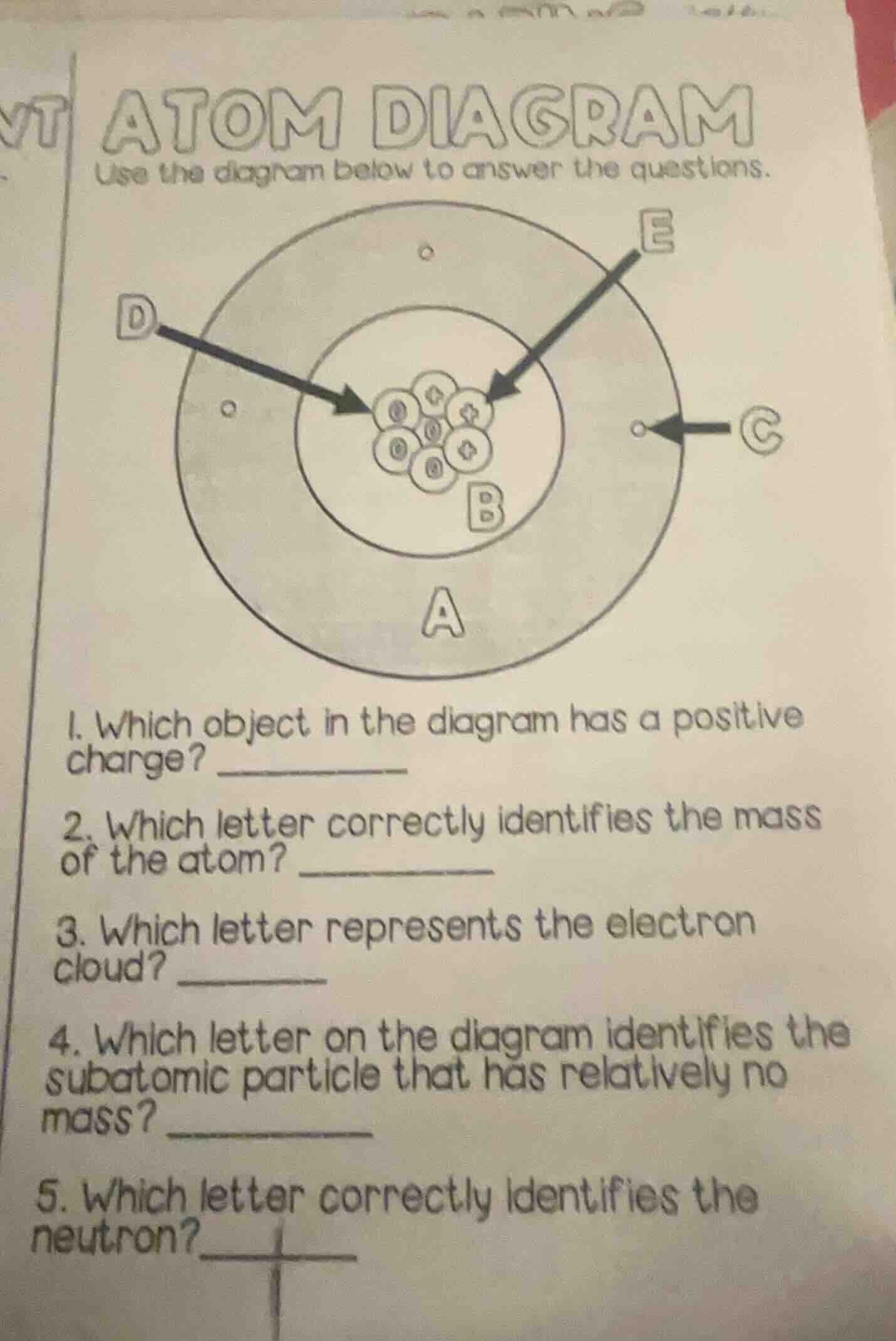
atom diagram
use the diagram below to answer the questions.
- which object in the diagram has a positive charge?
- which letter correctly identifies the mass of the atom?
- which letter represents the electron cloud?
- which letter on the diagram identifies the subatomic particle that has relatively no mass?
- which letter correctly identifies the neutron?
Step1: Match charge to particle
Protons (E) have a positive charge.
Step2: Locate atomic mass source
Most atomic mass is in the nucleus (B), holding protons/neutrons.
Step3: Identify electron cloud region
The outer area (A) is the electron cloud.
Step4: Find near-massless particle
Electrons (C) have negligible relative mass.
Step5: Locate neutral subatomic particle
Neutrons (D) are the neutral nucleus particles.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- E
- B
- A
- C
- D