QUESTION IMAGE
Question
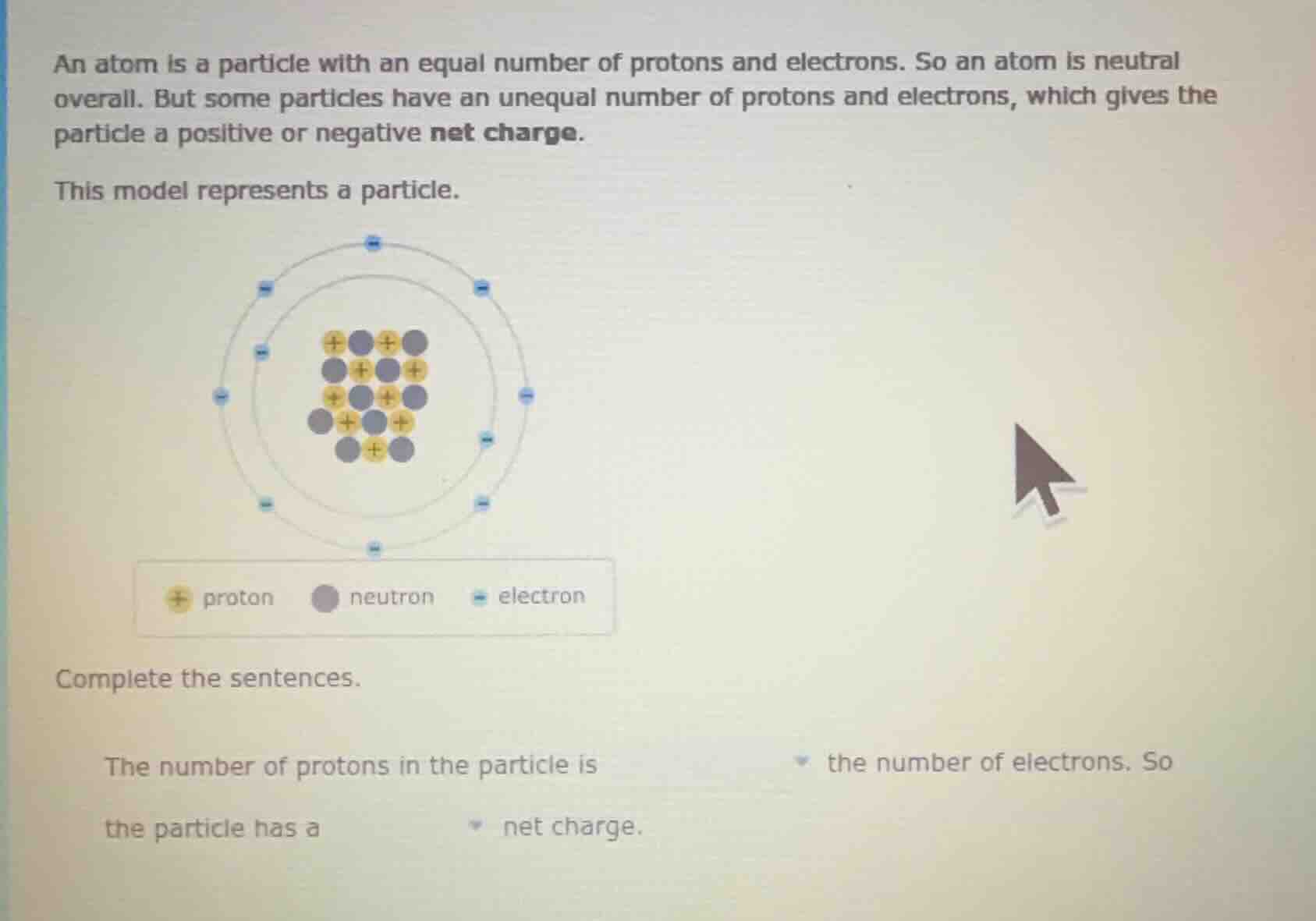
an atom is a particle with an equal number of protons and electrons. so an atom is neutral overall. but some particles have an unequal number of protons and electrons, which gives the particle a positive or negative net charge. this model represents a particle. image of atomic model with proton, neutron, electron legend complete the sentences. the number of protons in the particle is dropdown the number of electrons. so the particle has a dropdown net charge.
Step1: Count the number of protons
Looking at the model, the yellow '+' symbols (protons) are counted. Let's count them: In the nucleus, we can see the protons. Let's list them: First row: 2 (wait, no, let's count carefully. Let's see the nucleus: the yellow ones (protons) are: let's count each yellow circle. Let's do it step by step. The nucleus has protons: let's see, the first row (top) has two protons? Wait, no, let's look at the image. Wait, the user's image: the nucleus has protons (yellow with +) and neutrons (gray). Let's count protons: let's see, the protons are: let's count each yellow circle. Let's see:
Looking at the nucleus (the inner part), the protons (yellow) are: let's count:
First, let's list all yellow '+' (protons):
Row 1 (top): +, + (wait, no, the first row has +, gray, +, gray. So two protons.
Row 2: gray, +, gray, +. So two protons.
Row 3: +, gray, +, gray. So two protons.
Row 4: gray, +, gray, +. So two protons.
Row 5: +. Wait, no, maybe I'm miscounting. Wait, maybe a better way: let's count all yellow circles. Let's see:
Looking at the nucleus, the protons are: let's count each yellow '+' symbol. Let's do it:
First, the top part: +, + (wait, no, the first row (topmost) has +, gray, +, gray. So two protons.
Second row: gray, +, gray, +. Two protons.
Third row: +, gray, +, gray. Two protons.
Fourth row: gray, +, gray, +. Two protons.
Fifth row: gray, +, gray. Wait, no, maybe the nucleus has 9 protons? Wait, no, let's count again. Wait, maybe the correct count is: let's see the electrons first. The electrons (blue with -) are on the orbits. Let's count electrons: the outer orbit has 6 electrons? Wait, no, the orbits: the inner orbit (closer to nucleus) and outer orbit? Wait, the image shows two orbits? Wait, no, the electrons are on two rings? Wait, the blue electrons: let's count them. Let's see: the inner ring (closer to nucleus) has how many? Wait, no, the image: the electrons are on two circular paths. Let's count the blue electrons:
Looking at the image, the electrons (blue) are: let's count each blue circle. Let's see:
On the inner orbit (closer to nucleus): wait, no, maybe there are two orbits. Wait, the first (inner) orbit: how many? Wait, no, the image shows:
Wait, the electrons are on two circles? Wait, no, let's count:
Looking at the blue electrons:
First, the inner circle (closer to nucleus) has 4? No, wait, the image: let's count all blue electrons. Let's see:
Top: 1
Right (outer): 1
Bottom (outer): 1
Left (outer): 1
Then, inner circle: top (inner): 1, right (inner): 1, bottom (inner): 1, left (inner): 1? Wait, no, maybe I'm wrong. Wait, the image: the electrons are on two rings. Let's count:
Outer ring (larger circle): 6 electrons? Wait, no, let's count the blue dots:
Looking at the image, the blue electrons (electrons) are:
Top: 1
Right (outer): 1
Bottom (outer): 1
Left (outer): 1
Then, inner ring (smaller circle):
Top (inner): 1
Right (inner): 1
Bottom (inner): 1
Left (inner): 1
Wait, no, that's 8? Wait, no, let's count again. Wait, the image:
The electrons are on two concentric circles. Let's count each blue dot:
On the outer circle (larger radius):
- Top: 1
- Right (outer): 1
- Bottom (outer): 1
- Left (outer): 1
- Then, between top and right: 1
- Between right and bottom: 1
- Between bottom and left: 1
- Between left and top: 1? Wait, no, the image shows:
Wait, the original image (as per the user's problem) has the electron orbits: let's see, the electrons (blue) are:
Looking at the diagram, the electrons are on two rings. Let's count the numbe…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The number of protons in the particle is \boxed{greater than} the number of electrons. So the particle has a \boxed{positive} net charge.