QUESTION IMAGE
Question
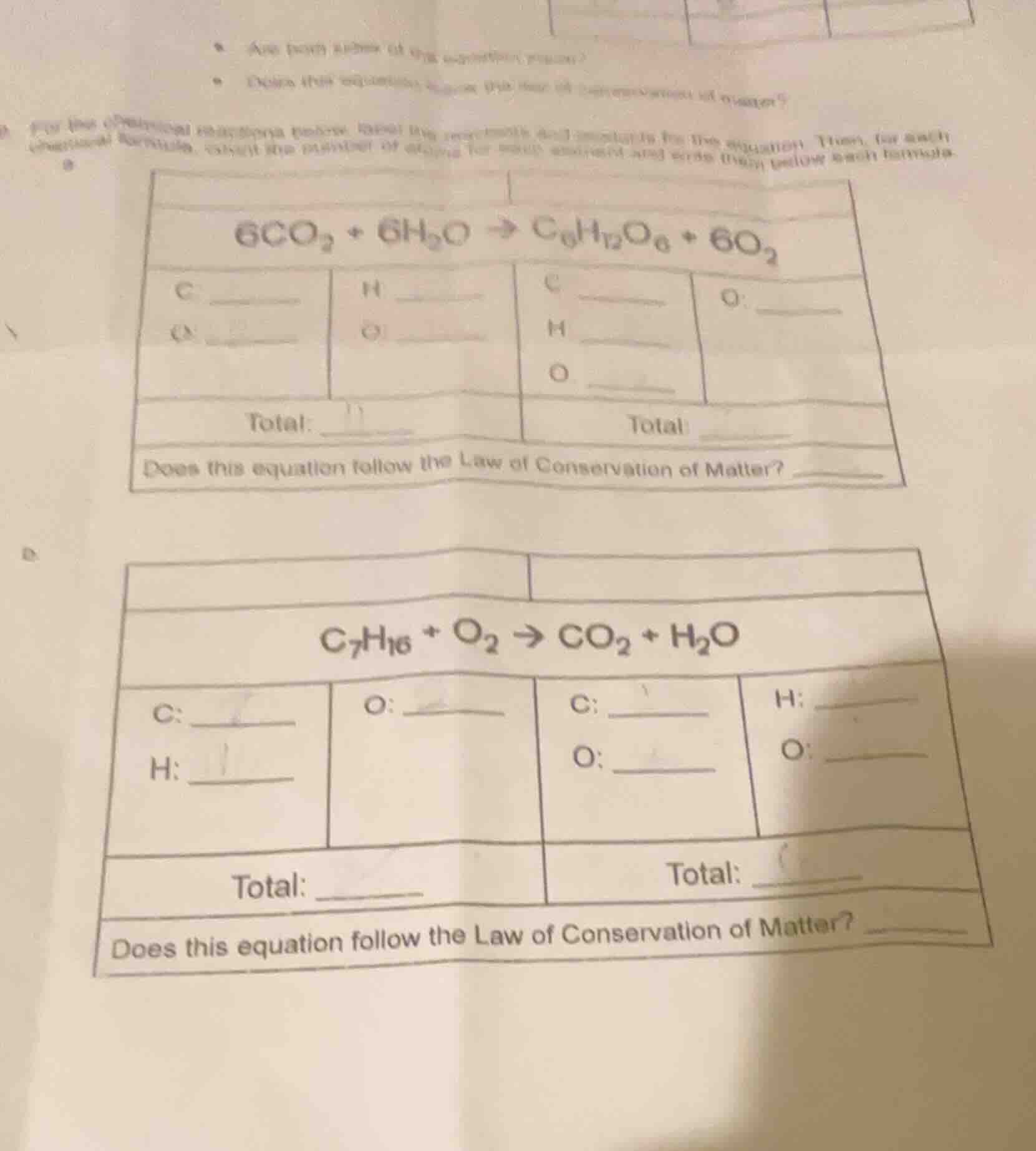
for the following reactions, recall the concepts for balancing equations. then, for each chemical formula, count the number of atoms for each element and write them below each formula.
part a
| reactants (left side) | products (right side) | ||
|---|---|---|---|
| o: ______ | o: ______ | h: ______ | |
| o: ______ | |||
| total: ______ | total: ______ |
does this equation follow the law of conservation of matter? ______
part b
| reactants (left side) | products (right side) | ||
|---|---|---|---|
| h: ______ | o: ______ | o: ______ | |
| total: ______ | total: ______ |
does this equation follow the law of conservation of matter? ______
Step1: Count reactant atoms (a)
For $\boldsymbol{6CO_2}$:
C: $6 \times 1 = 6$; O: $6 \times 2 = 12$
For $\boldsymbol{6H_2O}$:
H: $6 \times 2 = 12$; O: $6 \times 1 = 6$
Reactant totals:
C: 6; H: 12; O: $12 + 6 = 18$
Total atoms: $6 + 12 + 18 = 36$
Step2: Count product atoms (a)
For $\boldsymbol{C_6H_{12}O_6}$:
C: 6; H: 12; O: 6
For $\boldsymbol{6O_2}$:
O: $6 \times 2 = 12$
Product totals:
C: 6; H: 12; O: $6 + 12 = 18$
Total atoms: $6 + 12 + 18 = 36$
Step3: Verify Law (a)
Reactant total = Product total, so the law holds.
Step4: Count reactant atoms (b)
For $\boldsymbol{C_7H_{16}}$:
C: 7; H: 16
For $\boldsymbol{O_2}$:
O: 2
Reactant totals:
C:7; H:16; O:2
Total atoms: $7 + 16 + 2 = 25$
Step5: Count product atoms (b)
For $\boldsymbol{CO_2}$:
C:1; O:2
For $\boldsymbol{H_2O}$:
H:2; O:1
Product totals:
C:1; H:2; O: $2 + 1 = 3$
Total atoms: $1 + 2 + 3 = 6$
Step6: Verify Law (b)
Reactant total ≠ Product total, so the law does not hold.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Part a:
| Reactants | Products | ||||||
|---|---|---|---|---|---|---|---|
| $\boldsymbol{C_6H_{12}O_6}$ | C: 6 | H: 12 | O: 6 | $\boldsymbol{6O_2}$ | O: 12 | ||
| Total: | 36 | Total: | 36 | ||||
| Does this equation follow the Law of Conservation of Matter? | Yes |
Part b:
| Reactants | Products | ||||||
|---|---|---|---|---|---|---|---|
| $\boldsymbol{CO_2}$ | C: 1 | O: 2 | $\boldsymbol{H_2O}$ | H: 2 | O: 1 | ||
| Total: | 25 | Total: | 6 | ||||
| Does this equation follow the Law of Conservation of Matter? | No |