QUESTION IMAGE
Question
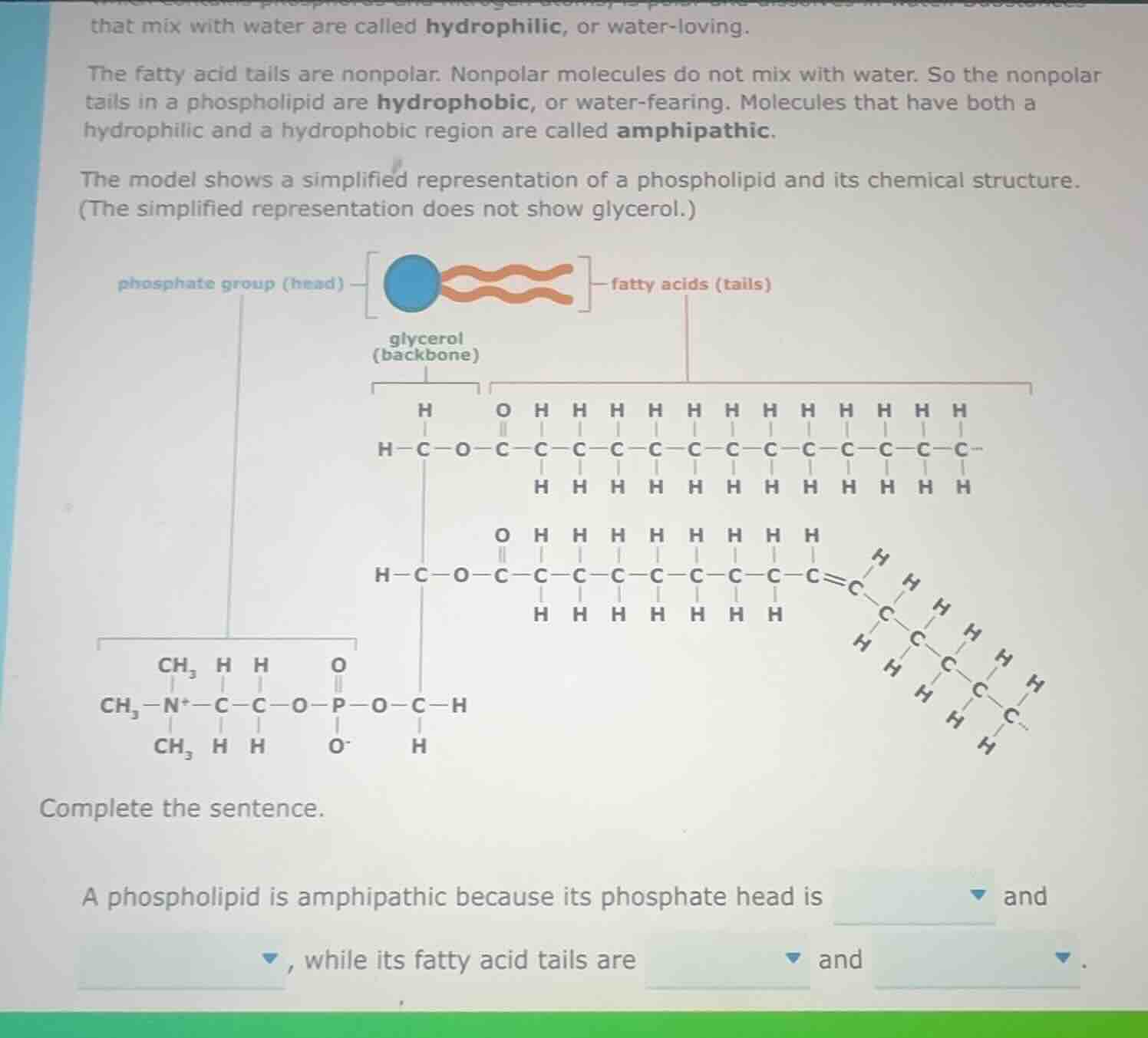
that mix with water are called hydrophilic, or water-loving.
the fatty acid tails are nonpolar. nonpolar molecules do not mix with water. so the nonpolar
tails in a phospholipid are hydrophobic, or water-fearing. molecules that have both a
hydrophilic and a hydrophobic region are called amphipathic.
the model shows a simplified representation of a phospholipid and its chemical structure.
(the simplified representation does not show glycerol.)
diagram of phospholipid structure
complete the sentence.
a phospholipid is amphipathic because its phosphate head is dropdown and
dropdown, while its fatty acid tails are dropdown and dropdown.
From the text, we know:
- Hydrophilic molecules mix with water. The phosphate head (polar region) is hydrophilic (water - loving) and polar (due to the phosphate group's nature).
- Hydrophobic molecules don't mix with water. The fatty acid tails are nonpolar and hydrophobic (water - fearing) as they are nonpolar.
So we fill in the blanks based on these definitions.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A phospholipid is amphipathic because its phosphate head is \(\boldsymbol{\text{hydrophilic}}\) and \(\boldsymbol{\text{polar}}\), while its fatty acid tails are \(\boldsymbol{\text{hydrophobic}}\) and \(\boldsymbol{\text{nonpolar}}\).