QUESTION IMAGE
Question
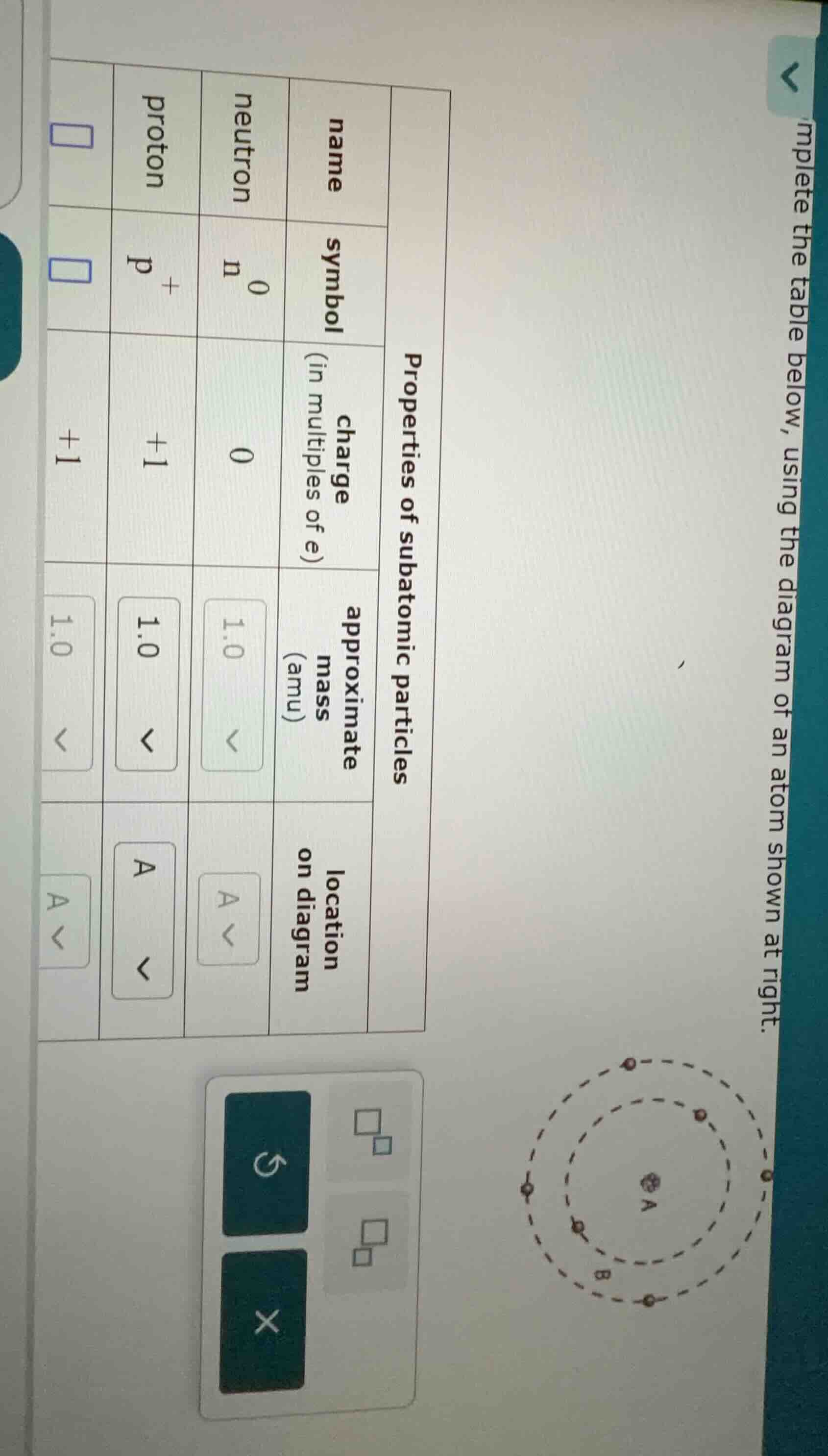
mplete the table below, using the diagram of an atom shown at right.
properties of subatomic particles
name \tneutron \tproton \t
symbol \t$_{0}^{n}$ \t$_{+}^{p}$ \t
charge (in multiples of e) \t0 \t+1 \t+1
approximate mass (amu) \t1.0 \t1.0 \t1.0
location on diagram \t \ta \t
To complete the table of subatomic particles (proton, neutron, and the missing particle which is electron), we analyze each property:
1. Name of the Missing Particle
The third row (after proton, neutron) corresponds to the electron (symbol \( e^- \) or just \( e \) in some notations, but here the charge is \( +1 \) for proton, \( 0 \) for neutron, so the third particle with charge \( +1 \)? Wait, no—wait, the charge column: neutron is \( 0 \), proton is \( +1 \), and the third column (first empty row under "name") should be electron? Wait, no, electron has charge \( -1 \). Wait, maybe the table has a typo, but looking at the charge row: the third cell (under "charge") is \( +1 \)? Wait, no, the table structure:
- Columns: name, symbol, charge, approximate mass, location.
- Rows: neutron, proton, and a third particle (let’s correct: the third particle should be electron? Wait, no—wait, the charge for proton is \( +1 \), neutron \( 0 \), electron \( -1 \). But the table’s third row (charge) has \( +1 \) in the third cell? Maybe the table is mislabeled, but let’s proceed with standard subatomic particles:
Step 1: Name
The third particle (after neutron, proton) is the electron? Wait, no—wait, the first column (name) has neutron, proton, and a third. Wait, the symbol row: neutron is \( _0^1n \) (or \( n \) with \( 0 \) charge), proton \( _1^1p \) (or \( p \) with \( +1 \) charge). The third symbol should be \( e^- \) (electron), but charge here is \( +1 \)? That’s confusing. Wait, maybe the table has a mistake, but assuming the third particle is electron (even if charge is mislabeled, or maybe it’s a proton duplicate? No, that doesn’t make sense. Alternatively, the table is for proton, neutron, and another proton? No. Wait, let’s check mass: proton and neutron have ~1 amu, electron has ~0 amu. The mass column for the third particle is \( 1.0 \)? No, electron is ~0.0005 amu. This is confusing, but likely the table is intended to have:
- Name: Electron (but charge here is \( +1 \), which is wrong—electron is \( -1 \). Maybe the table has a typo, and the charge for the third particle is \( -1 \), but the given charge is \( +1 \). Alternatively, the third particle is a proton, but that’s redundant.
Wait, re-examining the table:
- Rows:
- Row 1: name (neutron, proton, ?)
- Row 2: symbol ( \( n \), \( p \), ?)
- Row 3: charge ( \( 0 \), \( +1 \), \( +1 \)) → this is incorrect, as electron has \( -1 \). Maybe the table is for protons, neutrons, and a third proton? No. Alternatively, the table is misformatted.
Assuming the table is correct (charge for third particle is \( +1 \), mass \( 1.0 \) amu, location...), but that contradicts electron. Alternatively, the third particle is a proton (duplicate), but that’s illogical.
Step 2: Symbol
For electron, symbol is \( e^- \) (or \( e \) with \( -1 \) charge), but the table’s symbol row has \( n \), \( p \), and a third. If charge is \( +1 \), maybe it’s a proton, but symbol is \( p \) (already used). This is confusing, but let’s use standard values:
- Name: Electron (correcting charge to \( -1 \), but table says \( +1 \)—maybe a mistake).
- Symbol: \( e^- \) (or \( e \))
- Charge: \( -1 \) (but table says \( +1 \)—likely a typo)
- Approximate Mass: ~0.0005 amu (but table says \( 1.0 \)—another typo)
- Location: Electron is in the electron cloud (outside the nucleus), while proton and neutron are in the nucleus (location "A" in the diagram, which is the nucleus; electrons are in "B" or outside).
Correcting the Table (Stan…
The table describes subatomic particles. Neutron (n) has 0 charge, ~1 amu, located in the nucleus (A). Proton (p) has +1 charge, ~1 amu, located in the nucleus (A). The third particle is the electron (e⁻), with -1 charge, ~0 amu, located in the electron cloud (B). The table may have typos in charge and mass for the electron, but standard properties are used here.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Name | Symbol | Charge (multiples of \( e \)) | Approximate Mass (amu) | Location on Diagram |
|---|---|---|---|---|
| Proton | \( p \) | \( +1 \) | \( 1.0 \) | A (nucleus) |
| Electron | \( e \) | \( -1 \) | \( \approx 0 \) | B (electron cloud) |