QUESTION IMAGE
Question
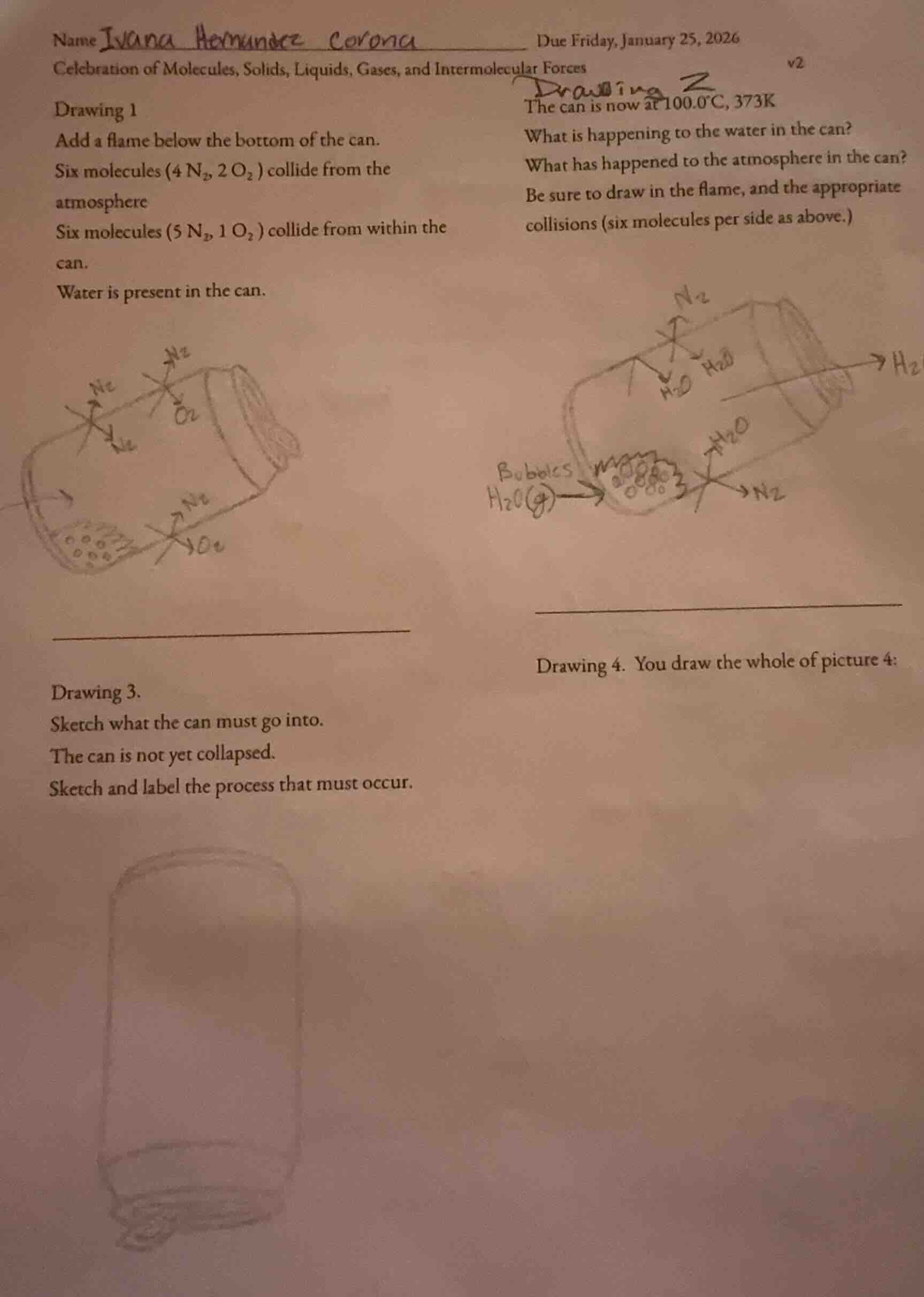
name ivana hernandez corona due friday, january 25, 2026 celebration of molecules, solids, liquids, gases, and intermolecular forces v2 drawing 1 add a flame below the bottom of the can. six molecules (4 n₂, 2 o₂) collide from the atmosphere six molecules (5 n₂, 1 o₂) collide from within the can. water is present in the can. drawing 2 the can is now at 100.0°c, 373k what is happening to the water in the can? what has happened to the atmosphere in the can? be sure to draw in the flame, and the appropriate collisions (six molecules per side as above.) drawing 3. sketch what the can must go into. the can is not yet collapsed. sketch and label the process that must occur. drawing 4. you draw the whole of picture 4:
For Drawing 2 Questions:
1. What is happening to the water in the can?
At 100.0°C (373K), water reaches its boiling point. Boiling occurs when the vapor pressure of water equals the atmospheric pressure. So the liquid water in the can is undergoing a phase change to water vapor (gas) as it boils. The heat from the flame provides the energy for the liquid water molecules to gain enough kinetic energy to escape the liquid phase and become gaseous \( \ce{H_2O} \) molecules, which is why bubbles (water vapor) form.
Initially, the can has an atmosphere with \( \ce{N_2} \) and \( \ce{O_2} \) molecules. When the water boils, the water vapor (\( \ce{H_2O(g)} \)) is produced. Also, the heating (from the flame) increases the kinetic energy of the gas molecules (both the original \( \ce{N_2} \), \( \ce{O_2} \), and the newly formed \( \ce{H_2O(g)} \)) in the can. This increased kinetic energy leads to more frequent and forceful collisions of the gas molecules with the walls of the can and with each other. Additionally, the water vapor adds to the total number of gas molecules in the can's atmosphere, increasing the total pressure (vapor pressure from water plus pressure from air molecules) inside the can.
To cause the can to collapse later (as part of the typical "collapsing can" experiment), the can must be placed in a cooler environment (like a beaker of cold water or ice water) after heating. The process that must occur is the cooling of the can and the water vapor inside it. When the can is moved to the cold environment (e.g., a beaker of cold water), the water vapor (\( \ce{H_2O(g)} \)) inside the can will cool down and condense back into liquid water (\( \ce{H_2O(l)} \)). This condensation reduces the amount of gas inside the can, which in turn reduces the pressure inside the can (since there are fewer gas molecules exerting pressure). The external atmospheric pressure (from the air outside the can) is then greater than the internal pressure, which will eventually cause the can to collapse (but in Drawing 3, it's not yet collapsed, so we sketch the setup: the can is placed in a container of cold water, and label the condensation of \( \ce{H_2O(g)} \) to \( \ce{H_2O(l)} \) and the resulting pressure difference).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The water in the can is boiling (undergoing vaporization), changing from liquid water (\( \ce{H_2O(l)} \)) to water vapor (\( \ce{H_2O(g)} \)) as it reaches its boiling point at 100.0°C (373K).