QUESTION IMAGE
Question
period: 5th date: 11/16/26
lesson 6: virtual urchin lab (part 2)
unit 2: homeostasis, assessment #1
page 21
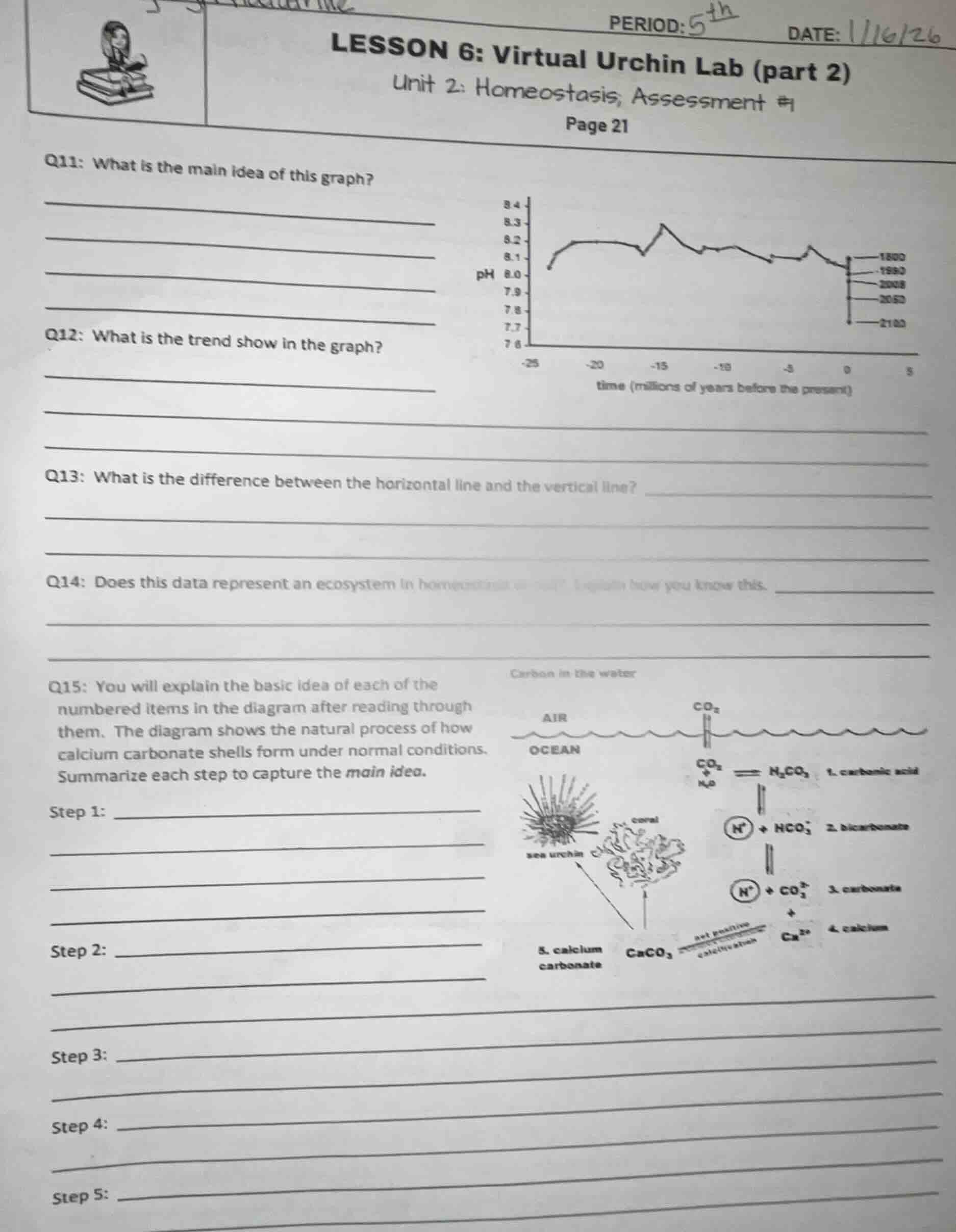
q11: what is the main idea of this graph?
q12: what is the trend show in the graph?
q13: what is the difference between the horizontal line and the vertical line?
q14: does this data represent an ecosystem in homeostasis or not? explain how you know this.
q15: you will explain the basic idea of each of the
numbered items in the diagram after reading through
them. the diagram shows the natural process of how
calcium carbonate shells form under normal conditions.
summarize each step to capture the main idea.
step 1:
step 2:
step 3:
step 4:
step 5:
(graph and diagram descriptions:
graph: ph vs time (millions of years before the present), with data points for 1800, 1990, 2008, 2050, 2100.
diagram: carbon in the water, showing process of calcium carbonate shell formation with steps: 1. carbonic acid, 2. bicarbonate, 3. carbonate, 4. calcium, 5. calcium carbonate, involving sea urchin, coral, co₂ from air to ocean, reactions with h₂o, na⁺, ca²⁺ etc.)
Q11: The graph tracks the change in ocean pH over millions of years, with markers for recent modern years to show current and projected pH levels.
Q12: Over most of the timeline, ocean pH stays relatively stable between ~8.0-8.3, but shows a sharp, accelerating downward (acidifying) trend as it approaches the present and future years.
Q13: The horizontal line (x-axis) represents time, measured in millions of years before the present. The vertical line (y-axis) represents the pH level of the ocean, a measure of acidity/alkalinity.
Q14: This data does NOT represent an ecosystem in homeostasis. Homeostasis requires stable, consistent conditions over time; the graph shows a recent, significant shift in ocean pH away from the long-term stable range, indicating a disruption of balance.
Q15:
Step 1: Atmospheric CO₂ dissolves in ocean water and reacts with H₂O to form carbonic acid (H₂CO₃).
Step 2: Carbonic acid breaks down into hydrogen ions (H⁺) and bicarbonate ions (HCO₃⁻).
Step 3: Bicarbonate breaks down further into more hydrogen ions and carbonate ions (CO₃²⁻).
Step 4: Dissolved calcium ions (Ca²⁺) are present in ocean water.
Step 5: Calcium ions combine with carbonate ions to form calcium carbonate (CaCO₃), which marine organisms like sea urchins and corals use to build their shells and skeletons.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Q11: It shows ocean pH changes over millions of years, including modern/future projections.
Q12: Ocean pH was stable for millions of years, but is now rapidly decreasing (ocean acidification) toward the present and future.
Q13: Horizontal line = time (millions of years before present); Vertical line = ocean pH level.
Q14: No, it does not. The recent sharp drop in pH breaks the long-term stable conditions required for homeostasis.
Q15:
Step 1: CO₂ from air forms carbonic acid in ocean water.
Step 2: Carbonic acid splits into H⁺ and bicarbonate.
Step 3: Bicarbonate splits into H⁺ and carbonate.
Step 4: Calcium ions are available in ocean water.
Step 5: Ca²⁺ and carbonate form calcium carbonate for marine organism shells/skeletons.