QUESTION IMAGE
Question
practice: atomic calculations
part 1 atomic structure
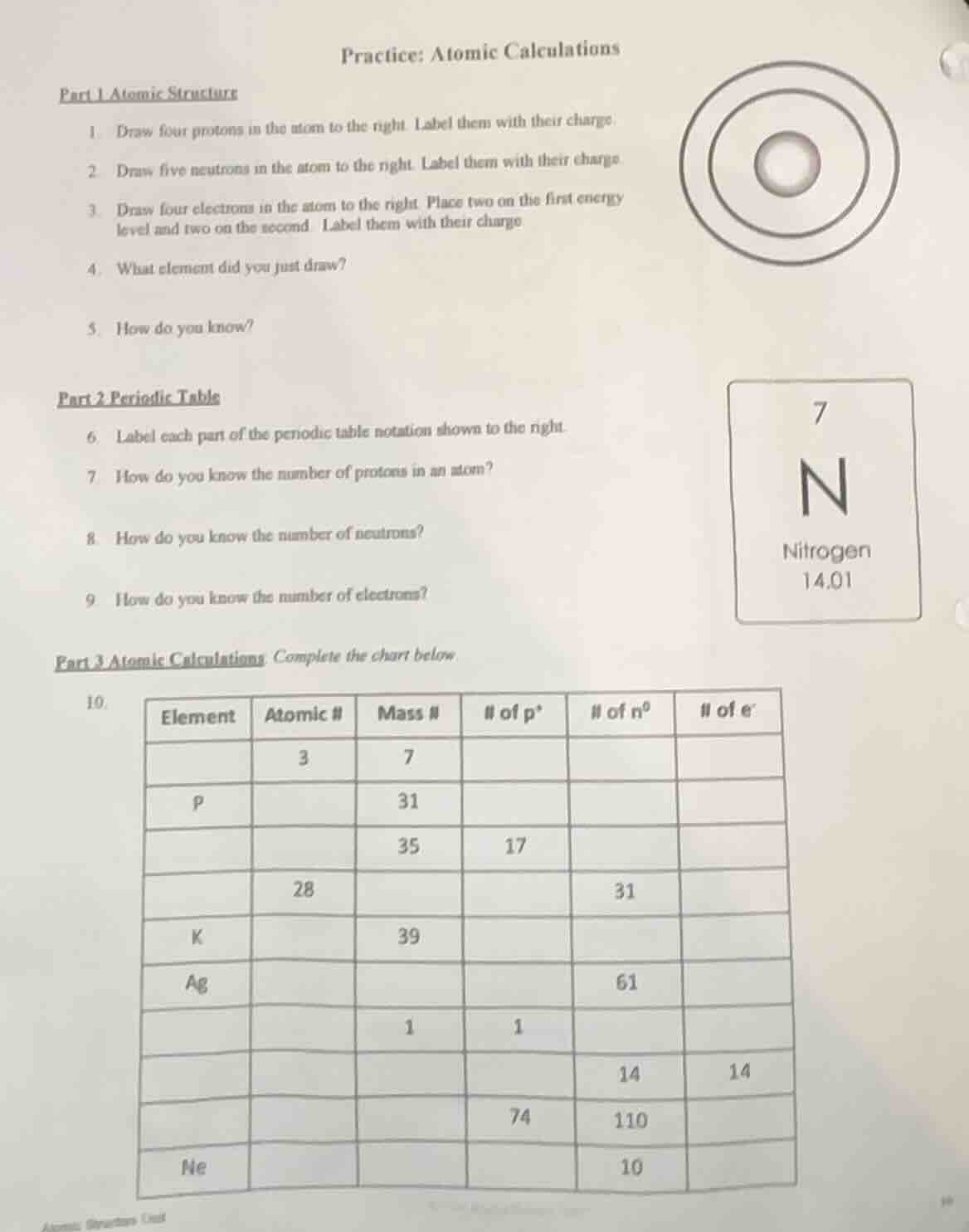
1 draw four protons in the atom to the right. label them with their charge.
2 draw five neutrons in the atom to the right. label them with their charge.
3 draw four electrons in the atom to the right. place two on the first energy
level and two on the second. label them with their charge
4 what element did you just draw?
5 how do you know?
part 2 periodic table
6 label each part of the periodic table notation shown to the right.
7 how do you know the number of protons in an atom?
8 how do you know the number of neutrons?
9 how do you know the number of electrons?
part 3 atomic calculations complete the chart below
10.
element atomic # mass # # of p⁺ # of n⁰ # of e⁻
3 7
p 31
35 17
28 31
k 39
ag 61
1 1
14 14
74 110
ne 10
Part 1: Atomic Structure
1. Drawing Protons
- Explanation: Protons are positively charged and located in the nucleus. We need to draw 4 protons in the nucleus (the central circle) and label them with \( + \).
- Action: In the central nucleus (the inner circle), draw 4 small circles or dots and label each with \( + \) (representing the positive charge of protons).
2. Drawing Neutrons
- Explanation: Neutrons are neutral (no charge) and also in the nucleus. We draw 5 neutrons in the nucleus and label them with \( 0 \) (since they have no charge).
- Action: In the central nucleus, draw 5 small circles or dots (along with the protons) and label each with \( 0 \) (representing the neutral charge of neutrons).
3. Drawing Electrons
- Explanation: Electrons are negatively charged and orbit the nucleus in energy levels. The first energy level (closest to the nucleus) can hold up to 2 electrons, and the second can hold up to 8 (but we have 2 here). We place 2 electrons on the first energy level (the inner electron shell) and 2 on the second (the outer electron shell) and label them with \( - \).
- Action: On the first energy level (the inner ring around the nucleus), draw 2 small circles or dots and label each with \( - \). On the second energy level (the outer ring), draw 2 small circles or dots and label each with \( - \).
4. Identifying the Element
- Explanation: The number of protons (atomic number) determines the element. We have 4 protons, so we look at the periodic table for the element with atomic number 4.
- Answer: Beryllium (Be)
5. Reasoning for the Element
- Explanation: The atomic number (number of protons) is unique to each element. Since we drew 4 protons, the atomic number is 4, which corresponds to Beryllium on the periodic table.
- Answer: The element is Beryllium because the number of protons (atomic number) is 4, and the element with atomic number 4 is Beryllium.
Part 2: Periodic Table
6. Labeling Periodic Table Notation (for Nitrogen, \( \ce{N} \) with atomic number 7, mass number ~14.01)
- Explanation: The periodic table notation has several parts: the atomic number (top left), the element symbol (middle), the element name (bottom), and the atomic mass (bottom right).
- Labels:
- Top number (7): Atomic number (number of protons)
- Middle letter (N): Element symbol
- Bottom word (Nitrogen): Element name
- Bottom number (14.01): Atomic mass (average mass of the element's isotopes)
7. Number of Protons in an Atom
- Explanation: The atomic number (the number at the top of the element's box on the periodic table) is equal to the number of protons in an atom of that element.
- Answer: The number of protons is equal to the atomic number (the top number in the element's box on the periodic table).
8. Number of Neutrons in an Atom
- Explanation: The mass number (rounded atomic mass) minus the atomic number gives the number of neutrons (since mass number = protons + neutrons).
- Formula: \( \text{Number of neutrons} = \text{Mass number} - \text{Atomic number} \)
- Answer: To find the number of neutrons, subtract the atomic number (number of protons) from the mass number (rounded atomic mass). So, \( \text{Neutrons} = \text{Mass number} - \text{Atomic number} \).
9. Number of Electrons in an Atom (for a neutral atom)
- Explanation: In a neutral atom, the number of electrons is equal to the number of protons (to balance the positive and negative charges).
- Answer: For a neutral atom, the num…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Part 1: Atomic Structure
1. Drawing Protons
- Explanation: Protons are positively charged and located in the nucleus. We need to draw 4 protons in the nucleus (the central circle) and label them with \( + \).
- Action: In the central nucleus (the inner circle), draw 4 small circles or dots and label each with \( + \) (representing the positive charge of protons).
2. Drawing Neutrons
- Explanation: Neutrons are neutral (no charge) and also in the nucleus. We draw 5 neutrons in the nucleus and label them with \( 0 \) (since they have no charge).
- Action: In the central nucleus, draw 5 small circles or dots (along with the protons) and label each with \( 0 \) (representing the neutral charge of neutrons).
3. Drawing Electrons
- Explanation: Electrons are negatively charged and orbit the nucleus in energy levels. The first energy level (closest to the nucleus) can hold up to 2 electrons, and the second can hold up to 8 (but we have 2 here). We place 2 electrons on the first energy level (the inner electron shell) and 2 on the second (the outer electron shell) and label them with \( - \).
- Action: On the first energy level (the inner ring around the nucleus), draw 2 small circles or dots and label each with \( - \). On the second energy level (the outer ring), draw 2 small circles or dots and label each with \( - \).
4. Identifying the Element
- Explanation: The number of protons (atomic number) determines the element. We have 4 protons, so we look at the periodic table for the element with atomic number 4.
- Answer: Beryllium (Be)
5. Reasoning for the Element
- Explanation: The atomic number (number of protons) is unique to each element. Since we drew 4 protons, the atomic number is 4, which corresponds to Beryllium on the periodic table.
- Answer: The element is Beryllium because the number of protons (atomic number) is 4, and the element with atomic number 4 is Beryllium.
Part 2: Periodic Table
6. Labeling Periodic Table Notation (for Nitrogen, \( \ce{N} \) with atomic number 7, mass number ~14.01)
- Explanation: The periodic table notation has several parts: the atomic number (top left), the element symbol (middle), the element name (bottom), and the atomic mass (bottom right).
- Labels:
- Top number (7): Atomic number (number of protons)
- Middle letter (N): Element symbol
- Bottom word (Nitrogen): Element name
- Bottom number (14.01): Atomic mass (average mass of the element's isotopes)
7. Number of Protons in an Atom
- Explanation: The atomic number (the number at the top of the element's box on the periodic table) is equal to the number of protons in an atom of that element.
- Answer: The number of protons is equal to the atomic number (the top number in the element's box on the periodic table).
8. Number of Neutrons in an Atom
- Explanation: The mass number (rounded atomic mass) minus the atomic number gives the number of neutrons (since mass number = protons + neutrons).
- Formula: \( \text{Number of neutrons} = \text{Mass number} - \text{Atomic number} \)
- Answer: To find the number of neutrons, subtract the atomic number (number of protons) from the mass number (rounded atomic mass). So, \( \text{Neutrons} = \text{Mass number} - \text{Atomic number} \).
9. Number of Electrons in an Atom (for a neutral atom)
- Explanation: In a neutral atom, the number of electrons is equal to the number of protons (to balance the positive and negative charges).
- Answer: For a neutral atom, the number of electrons is equal to the number of protons (atomic number), so electrons = protons (atomic number).
Part 3: Atomic Calculations (Completing the Chart)
We use the following relationships:
- Atomic number (\( Z \)) = number of protons (\( p^+ \)) = number of electrons (\( e^- \)) (for neutral atoms)
- Mass number (\( A \)) = number of protons (\( p^+ \)) + number of neutrons (\( n^0 \))
So, \( n^0 = A - Z \)
Let's complete each row:
| Element | Atomic # (\( Z \)) | Mass # (\( A \)) | # of \( p^+ \) | # of \( n^0 \) (\( A - Z \)) | # of \( e^- \) (\( Z \)) |
|---|---|---|---|---|---|
| 2. P | 15 (Atomic # of P) | 31 | 15 | \( 31 - 15 = 16 \) | 15 |
| 3. | 17 (Atomic # of Cl) | 35 | 17 | \( 35 - 17 = 18 \) | 17 |
| 4. | 28 | \( 28 + 31 = 59 \) | 28 | 31 | 28 |
| 5. K | 19 (Atomic # of K) | 39 | 19 | \( 39 - 19 = 20 \) | 19 |
| 6. Ag | 47 (Atomic # of Ag) | \( 47 + 61 = 108 \) | 47 | 61 | 47 |
| 7. | 1 | 1 | 1 | \( 1 - 1 = 0 \) | 1 |
| 8. | 14 (Atomic # of Si) | \( 14 + 14 = 28 \) | 14 | 14 | 14 |
| 9. | 74 (Atomic # of W) | \( 74 + 110 = 184 \) | 74 | 110 | 74 |
| 10. Ne | 10 (Atomic # of Ne) | \( 10 + 10 = 20 \) | 10 | 10 | 10 |
Detailed Calculations for Each Row:
- First Row (Atomic # 3, Mass # 7):
- \( p^+ = 3 \) (atomic #)
- \( n^0 = 7 - 3 = 4 \)
- \( e^- = 3 \) (neutral atom, so \( e^- = p^+ \))
- Phosphorus (P):
- Atomic # of P is 15, so \( p^+ = 15 \)
- \( n^0 = 31 - 15 = 16 \)
- \( e^- = 15 \)
- Chlorine (Cl, \( p^+ = 17 \)):
- Atomic # = 17, so \( Z = 17 \)
- \( n^0 = 35 - 17 = 18 \)
- \( e^- = 17 \)
- Atomic # 28, \( n^0 = 31 \):
- \( A = 28 + 31 = 59 \)
- \( p^+ = 28 \)
- \( e^- = 28 \)
- Potassium (K):
- Atomic # of K is 19, so \( p^+ = 19 \)
- \( n^0 = 39 - 19 = 20 \)
- \( e^- = 19 \)
- Silver (Ag):
- Atomic # of Ag is 47, so \( p^+ = 47 \)
- \( A = 47 + 61 = 108 \)
- \( e^- = 47 \)
- Hydrogen (H, \( p^+ = 1 \), \( A = 1 \)):
- \( n^0 = 1 - 1 = 0 \)
- \( e^- = 1 \)
- Silicon (Si, \( e^- = 14 \), \( n^0 = 14 \)):
- \( p^+ = 14 \) (since \( e^- = p^+ \) in neutral atom)
- \( A = 14 + 14 = 28 \)
- Atomic # = 14
- Tungsten (W, \( p^+ = 74 \), \( n^0 = 110 \)):
- \( A = 74 + 110 = 184 \)
- \( e^- = 74 \) (neutral atom)
- Neon (Ne):
- Atomic # of Ne is 10, so \( p^+ = 10 \)
- \( A = 10 + 10 = 20 \)
- \( e^- = 10 \)