QUESTION IMAGE
Question
question 2
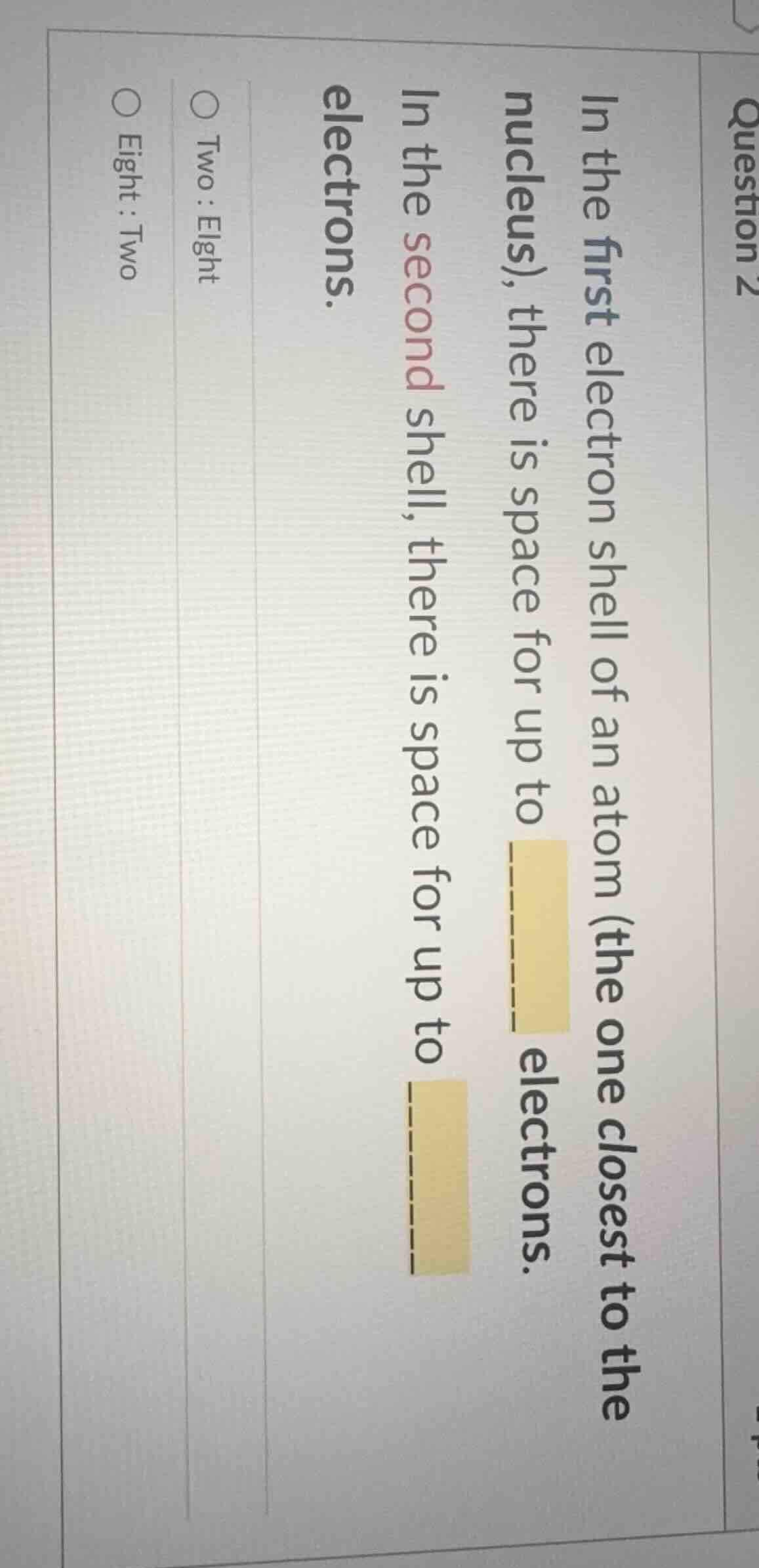
in the first electron shell of an atom (the one closest to the nucleus), there is space for up to ______ electrons.
in the second shell, there is space for up to ______ electrons.
○ two : eight
○ eight : two
Brief Explanations
The first electron shell (K shell) has a maximum capacity of 2 electrons, as it only has an s orbital. The second electron shell (L shell) has both s and p orbitals, giving it a maximum capacity of 8 electrons.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
○ Two : Eight