QUESTION IMAGE
Question
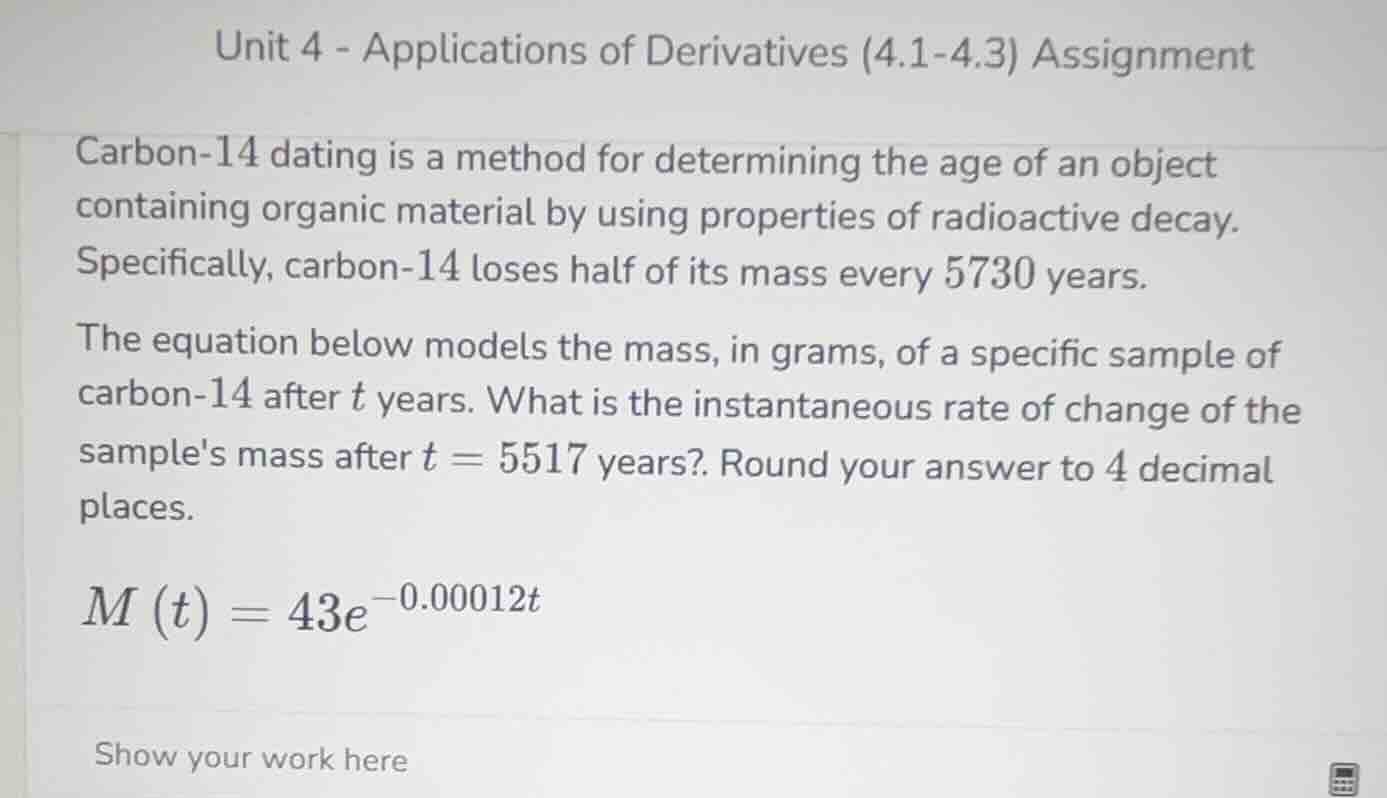
unit 4 - applications of derivatives (4.1-4.3) assignment
carbon-14 dating is a method for determining the age of an object containing organic material by using properties of radioactive decay. specifically, carbon-14 loses half of its mass every 5730 years.
the equation below models the mass, in grams, of a specific sample of carbon-14 after ( t ) years. what is the instantaneous rate of change of the samples mass after ( t = 5517 ) years? round your answer to 4 decimal places.
( m(t) = 43e^{-0.00012t} )
show your work here
Step1: Find derivative of $M(t)$
The derivative of $M(t)=43e^{-0.00012t}$ uses the chain rule: $\frac{d}{dt}[e^{kt}]=ke^{kt}$.
$\frac{dM}{dt} = 43 \times (-0.00012)e^{-0.00012t} = -0.00516e^{-0.00012t}$
Step2: Substitute $t=5517$
Calculate the exponent first: $-0.00012 \times 5517 = -0.66204$
$\frac{dM}{dt}\bigg|_{t=5517} = -0.00516e^{-0.66204}$
Step3: Compute the value
First calculate $e^{-0.66204} \approx 0.5150$, then multiply:
$-0.00516 \times 0.5150 \approx -0.002657$
Round to 4 decimal places: $-0.0027$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$-0.0027$ grams per year