QUESTION IMAGE
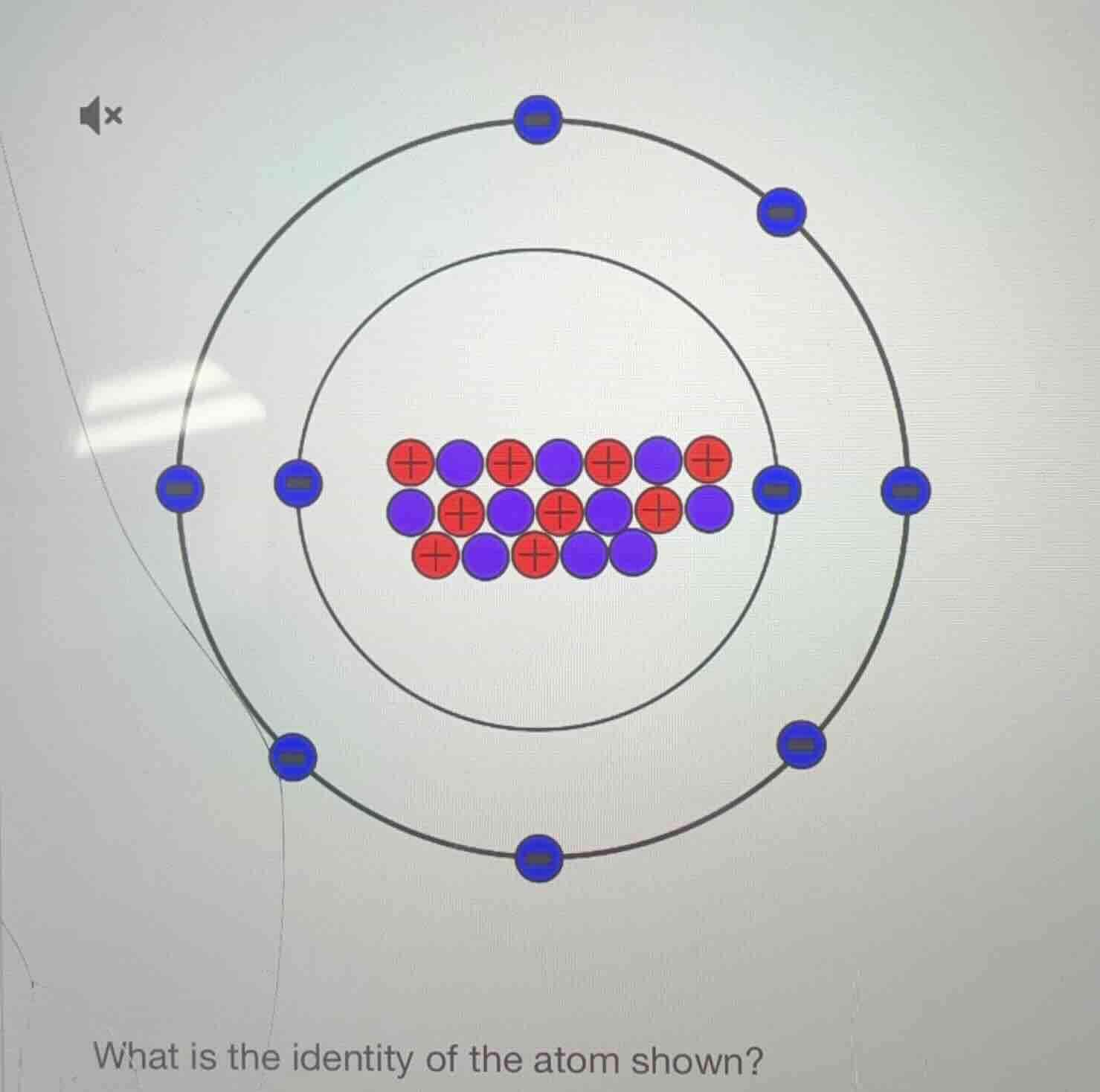
Question
what is the identity of the atom shown?
Step1: Count protons (red +)
Count red circles: Let's list them. First row: 4 (wait, no, let's count all red. First row: 4? Wait, looking at the nucleus: red (protons) and purple (neutrons). Let's count protons:
First row (top) of nucleus: 4 red? Wait, no, let's see:
Looking at the nucleus:
First row (horizontal top): red, purple, red, purple, red, purple, red → 4 red? Wait, no, first row (top) has 4 red? Wait, no, let's count all red:
First row (top): positions 1 (red), 3 (red), 5 (red), 7 (red) → 4? Wait, no, the nucleus:
First row (top layer): red, purple, red, purple, red, purple, red → 4 red? Wait, no, second row: purple, red, purple, red, purple, red, purple → 3 red? Wait, third row: red, purple, red, purple, purple → 2 red? Wait, no, maybe better to count:
Wait, the red circles (protons) in the nucleus: let's count each red:
First row (top) of nucleus: 4 red (positions 1,3,5,7? Wait, no, the first row (horizontal) has 7 circles? Wait, the nucleus has:
Top row (horizontal): 7 circles? Wait, no, the nucleus is a cluster. Let's count protons (red) and neutrons (purple).
Wait, the electron shells: inner shell (first energy level) has 2 electrons (blue -), outer shell (second) has 8? Wait, no, inner shell (the smaller circle) has 2 electrons (blue -), outer shell (larger) has 8? Wait, no, the inner electron shell (the middle circle) has 2 electrons, outer (the big circle) has 8? Wait, total electrons: 2 + 8 = 10? Wait, no, let's count electrons:
Inner electron shell (smaller orbit): 2 electrons (blue -). Outer electron shell (larger orbit): let's count the blue -: 8? Wait, the outer orbit has 8? Wait, the diagram: inner orbit (middle circle) has 2 electrons, outer orbit (big circle) has 8? Wait, no, let's count the blue -:
Inner orbit (middle circle): 2 electrons (left and right). Outer orbit (big circle): top, top-right, right, bottom-right, bottom, bottom-left, left, top-left? Wait, the outer orbit has 8 electrons? Wait, no, the image: inner orbit (middle circle) has 2 electrons (blue -), outer orbit (big circle) has 8? Wait, no, the inner orbit (the smaller circle) has 2 electrons, outer (the larger) has 8? Wait, total electrons: 2 + 8 = 10? Wait, but electrons equal protons (neutral atom). So protons should be 10.
Now count protons (red +):
In the nucleus, count red circles:
Let's list all red:
First row (top) of nucleus: 4 red? Wait, no, let's count:
Looking at the nucleus:
Row 1 (top): red, purple, red, purple, red, purple, red → 4 red.
Row 2 (middle): purple, red, purple, red, purple, red, purple → 3 red.
Row 3 (bottom): red, purple, red, purple, purple → 2 red.
Wait, 4 + 3 + 2 = 9? No, that can't be. Wait, maybe I miscounted. Let's do it again.
Alternative approach: electrons = protons (neutral atom). Let's count electrons:
Inner electron shell (smaller circle): 2 electrons (blue -).
Outer electron shell (larger circle): let's count the blue -: top (1), top-right (2), right (3), bottom-right (4), bottom (5), bottom-left (6), left (7), top-left (8). Wait, 8 electrons. So total electrons: 2 + 8 = 10. Therefore, protons = 10 (since neutral atom, protons = electrons).
Now count protons (red +) in the nucleus: let's count red circles.
Looking at the nucleus:
First row (top): red, purple, red, purple, red, purple, red → 4 red.
Second row (middle): purple, red, purple, red, purple, red, purple → 3 red.
Third row (bottom): red, purple, red, purple, purple → 2 red. Wait, 4 + 3 + 2 = 9. No, that's wrong. Wait, maybe the bottom row has 3? Wait, bottom row: red, purple, red, purple, purple → no, the botto…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Neon (or Ne)