QUESTION IMAGE
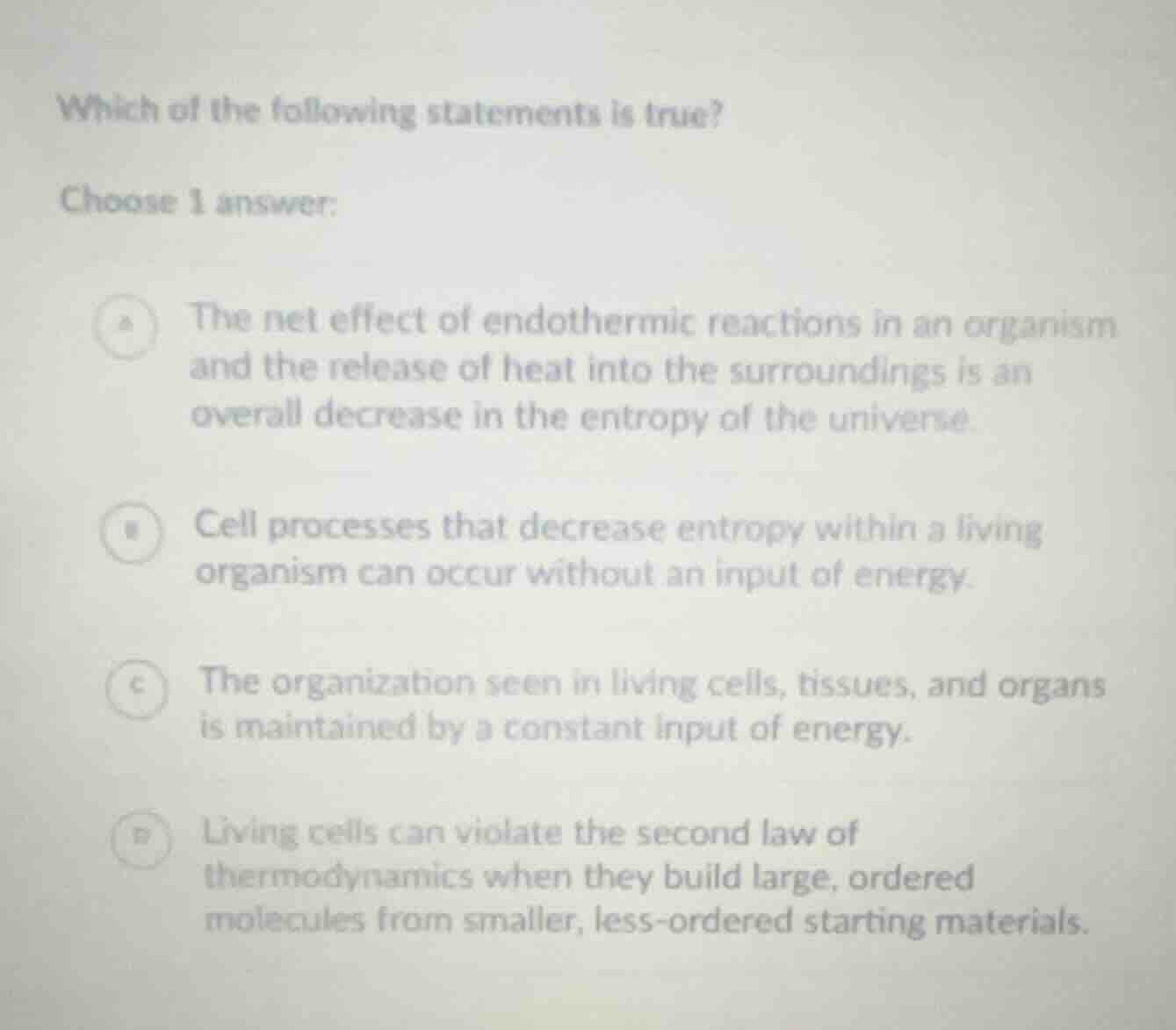
Question
which of the following statements is true?
choose 1 answer:
a the net effect of endothermic reactions in an organism and the release of heat into the surroundings is an overall decrease in the entropy of the universe.
b cell processes that decrease entropy within a living organism can occur without an input of energy.
c the organization seen in living cells, tissues, and organs is maintained by a constant input of energy.
d living cells can violate the second law of thermodynamics when they build large, ordered molecules from smaller, less - ordered starting materials.
- Option A: Violates the 2nd law of thermodynamics; the universe's entropy always increases overall, even with endothermic reactions and heat release.
- Option B: Decreasing entropy (increasing order) in cells requires energy input, as it is a non-spontaneous process.
- Option C: Living systems are open systems; they maintain high organization (low internal entropy) by constantly taking in energy (e.g., via food or sunlight) to drive processes, which aligns with thermodynamic laws.
- Option D: Living cells do not violate the 2nd law; while they create local order, the total entropy of the universe (cells + surroundings) increases.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. The organization seen in living cells, tissues, and organs is maintained by a constant input of energy.