QUESTION IMAGE
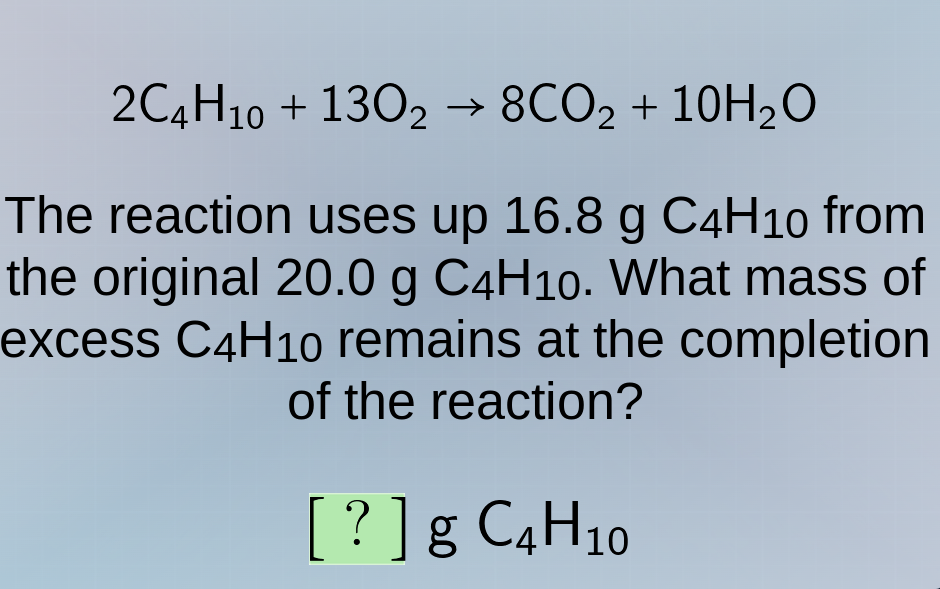
Question
2c₄h₁₀ + 13o₂ → 8co₂ + 10h₂o
the reaction uses up 16.8 g c₄h₁₀ from
the original 20.0 g c₄h₁₀. what mass of
excess c₄h₁₀ remains at the completion
of the reaction?
? g c₄h₁₀
Step1: Identify given values
Original mass of \( C_4H_{10} = 20.0 \, \text{g} \), Mass of \( C_4H_{10} \) used = \( 16.8 \, \text{g} \)
Step2: Calculate excess mass
Excess mass = Original mass - Mass used
\( = 20.0 - 16.8 \)
\( = 3.2 \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 3.2 \)