QUESTION IMAGE
Question
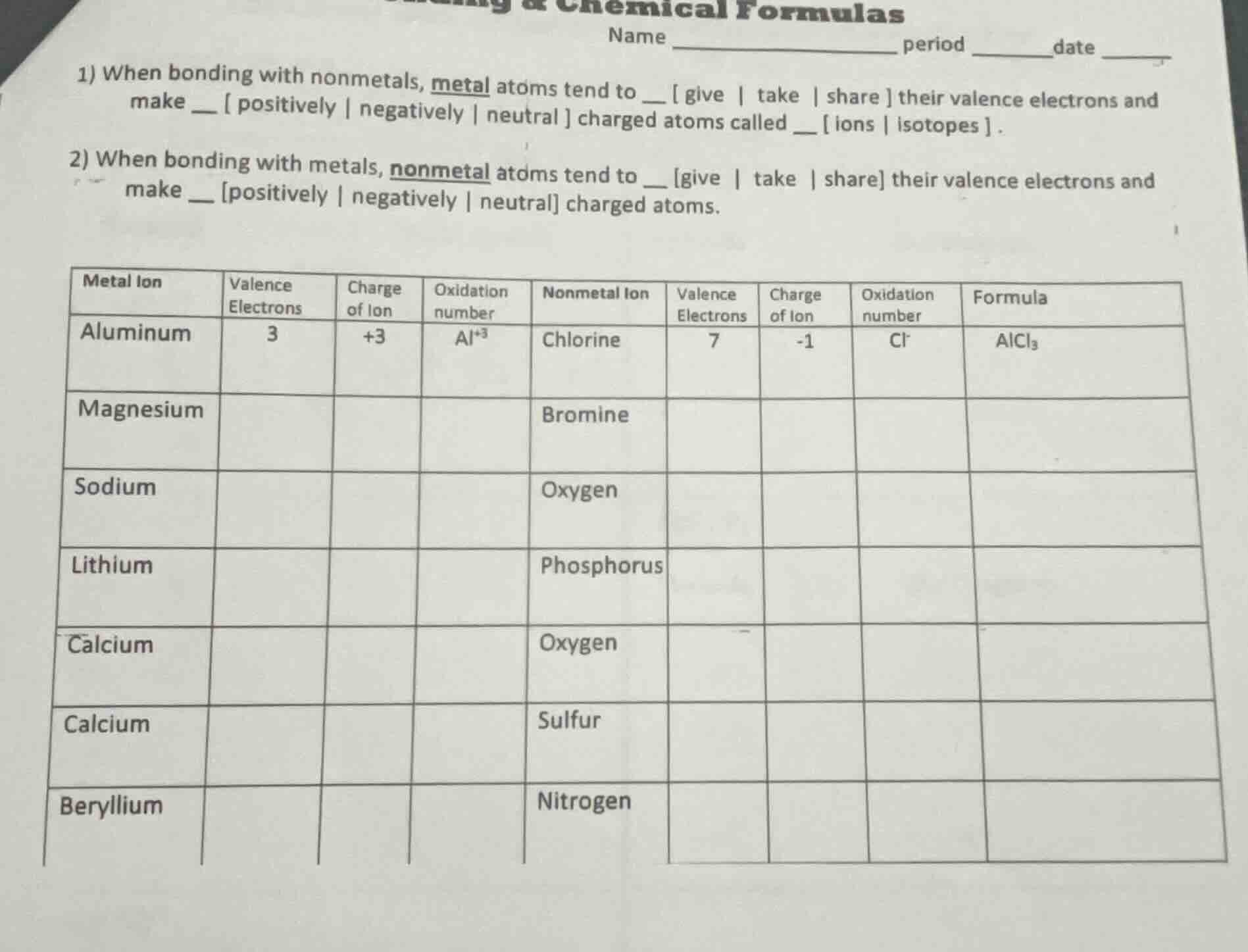
chemical formulas
name _____________ period __date ____
- when bonding with nonmetals, metal atoms tend to give | take | share their valence electrons and make positively | negatively | neutral charged atoms called __ ions | isotopes .
- when bonding with metals, nonmetal atoms tend to give | take | share their valence electrons and make positively | negatively | neutral charged atoms.
| metal ion | valence electrons | charge of ion | oxidation number | nonmetal ion | valence electrons | charge of ion | oxidation number | formula |
| aluminum | 3 | +3 | al⁺³ | chlorine | 7 | -1 | cl⁻ | alcl₃ |
| magnesium | bromine | |||||||
| sodium | oxygen | |||||||
| lithium | phosphorus | |||||||
| calcium | oxygen | |||||||
| calcium | sulfur | |||||||
| beryllium | nitrogen |
Question 1
Metals have few valence electrons (usually 1 - 3) and tend to lose (give) them to achieve a stable electron configuration. When they lose electrons, they have more protons than electrons, so they become positively charged. These charged atoms are called ions (isotopes are atoms of the same element with different neutrons, not related to charge from bonding).
Nonmetals have 5 - 7 valence electrons (usually) and tend to gain (take) electrons to achieve a stable octet. When they gain electrons, they have more electrons than protons, so they become negatively charged.
Step1: Determine Magnesium's valence electrons, charge, oxidation number
Magnesium is in group 2, so it has 2 valence electrons. It will lose 2 electrons, so charge of ion is \(+2\), oxidation number is \(Mg^{+2}\).
Step2: Determine Bromine's valence electrons, charge, oxidation number
Bromine is in group 17, has 7 valence electrons. It will gain 1 electron, so charge of ion is \(-1\), oxidation number is \(Br^{-}\).
Step3: Determine the formula
To form a neutral compound, the total positive charge equals total negative charge. Magnesium is \(+2\), Bromine is \(-1\). Let the number of Mg be \(x\) and Br be \(y\). So \(2x = 1y\). The simplest ratio is \(x = 1\), \(y = 2\), so formula is \(MgBr_2\).
[SSE onError error]
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
When bonding with nonmetals, metal atoms tend to \(\boldsymbol{\text{give}}\) their valence electrons and make \(\boldsymbol{\text{positively}}\) charged atoms called \(\boldsymbol{\text{ions}}\).