QUESTION IMAGE
Question
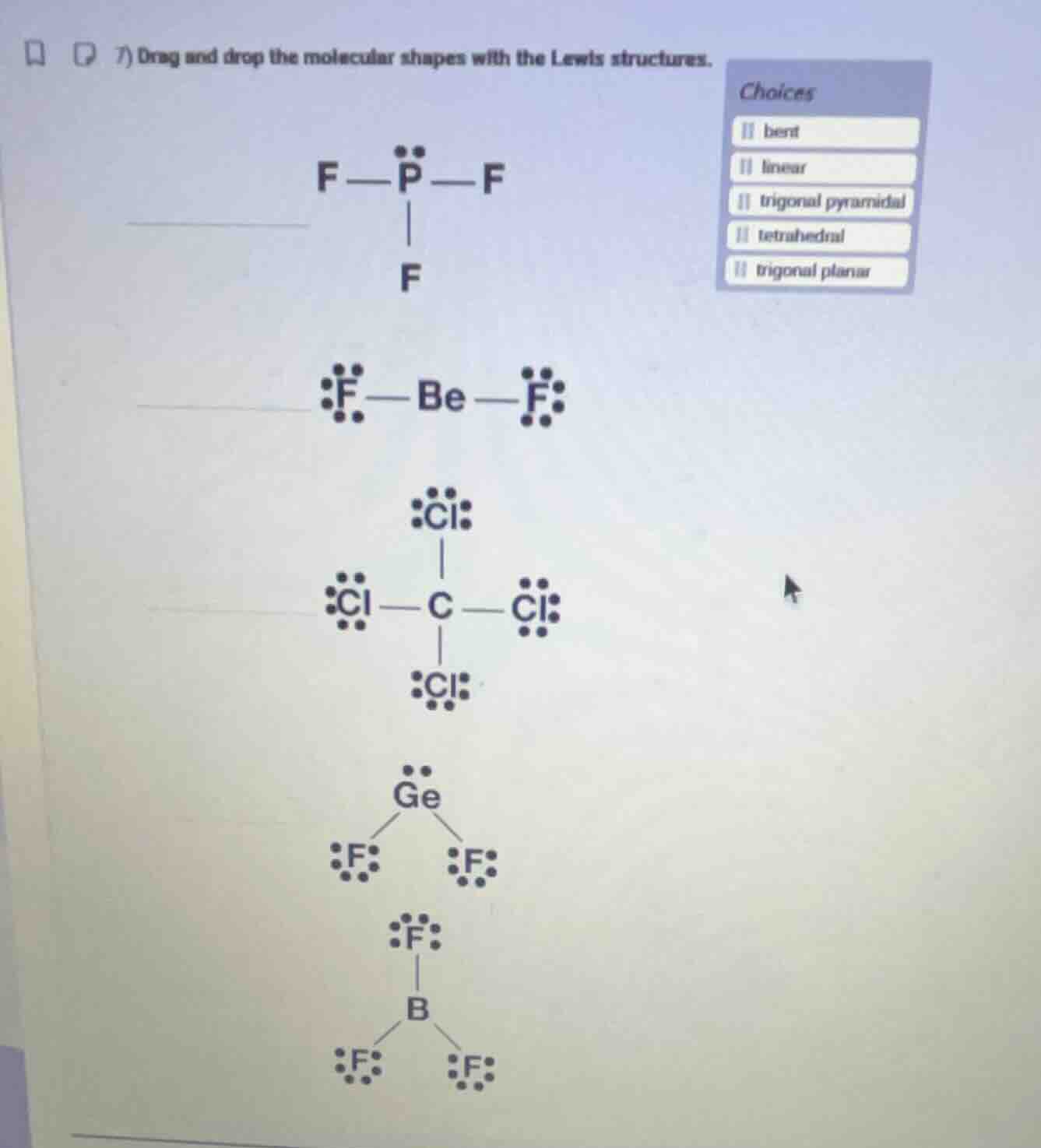
- drag and drop the molecular shapes with the lewis structures.
choices
bent
linear
trigonal pyramidal
tetrahedral
trigonal planar
Brief Explanations
- For $\text{PF}_3$: Central P has 3 bonding pairs + 1 lone pair, so electron repulsion forms a trigonal pyramidal shape.
- For $\text{BeF}_2$: Central Be has 2 bonding pairs, no lone pairs, so atoms align linearly.
- For $\text{CCl}_4$: Central C has 4 bonding pairs, no lone pairs, forming a tetrahedral shape.
- For $\text{GeF}_2$: Central Ge has 2 bonding pairs + 1 lone pair, lone pair repulsion creates a bent shape.
- For $\text{BF}_3$: Central B has 3 bonding pairs, no lone pairs, forming a trigonal planar shape.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{F-P-F (with one lone pair on P, three F atoms)}$: trigonal pyramidal
- $\text{F-Be-F (with three lone pairs on each F)}$: linear
- $\text{Cl-C-Cl (four Cl atoms around C)}$: tetrahedral
- $\text{F-Ge-F (with one lone pair on Ge, two F atoms)}$: bent
- $\text{F-B-F (three F atoms around B)}$: trigonal planar