QUESTION IMAGE
Question
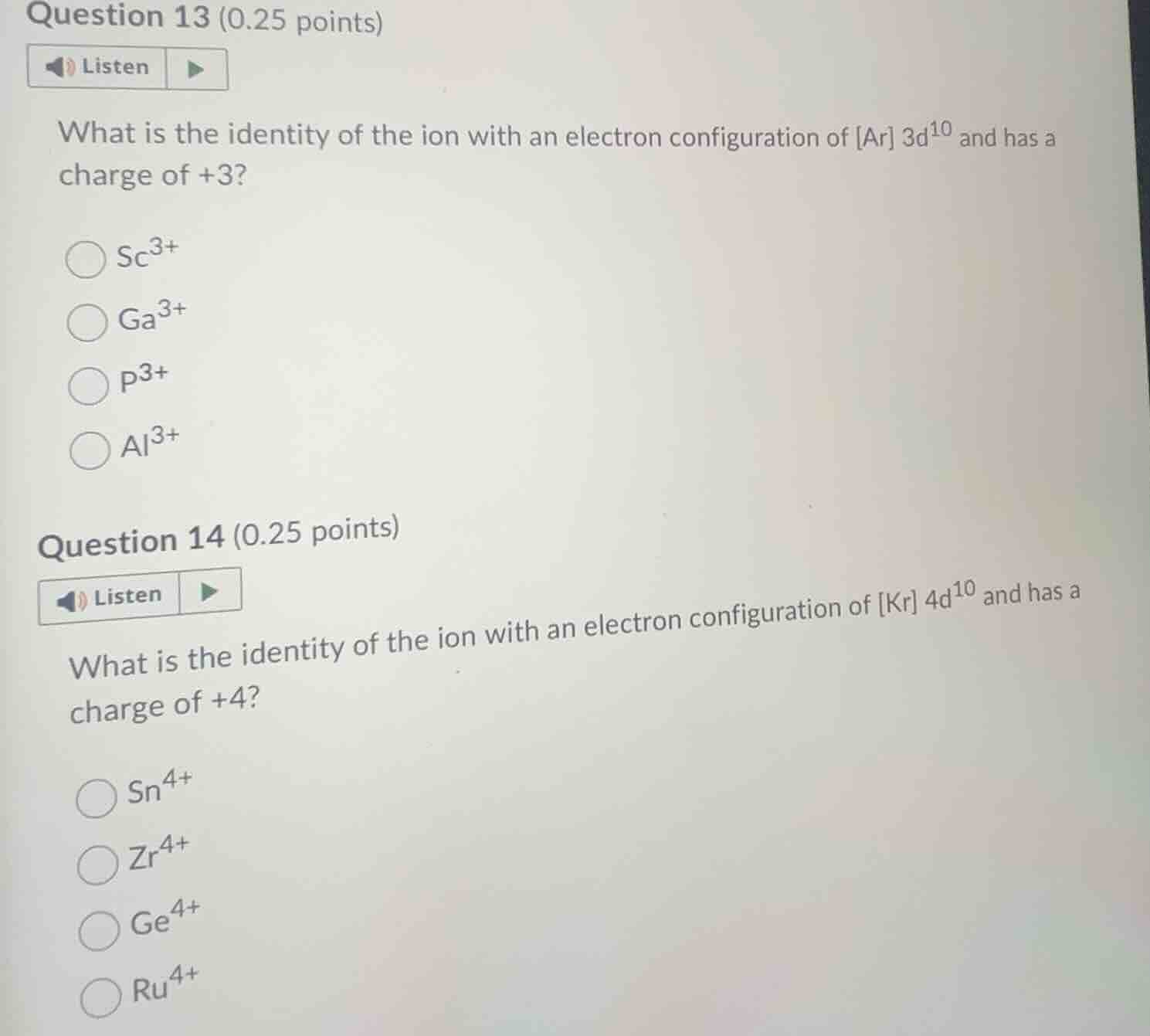
question 13 (0.25 points)listenwhat is the identity of the ion with an electron configuration of ar $3d^{10}$ and has a charge of +3?$ce{sc^{3+}}�LXB0�ce{p^{3+}}�LXB1�ce{zr^{4+}}�LXB2�ce{ru^{4+}}$
Step1: Find neutral atom e⁻ count (Q13)
Ion e⁻ count = Ar electrons + 10 = 18 + 10 = 28. Neutral atom e⁻ = 28 + 3 = 31.
Step2: Match atomic number (Q13)
Atomic number 31 corresponds to Ga, so ion is $\text{Ga}^{3+}$.
Step3: Find neutral atom e⁻ count (Q14)
Ion e⁻ count = Kr electrons + 10 = 36 + 10 = 46. Neutral atom e⁻ = 46 + 4 = 50.
Step4: Match atomic number (Q14)
Atomic number 50 corresponds to Sn, so ion is $\text{Sn}^{4+}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 13: B. $\text{Ga}^{3+}$
Question 14: A. $\text{Sn}^{4+}$