QUESTION IMAGE
Question
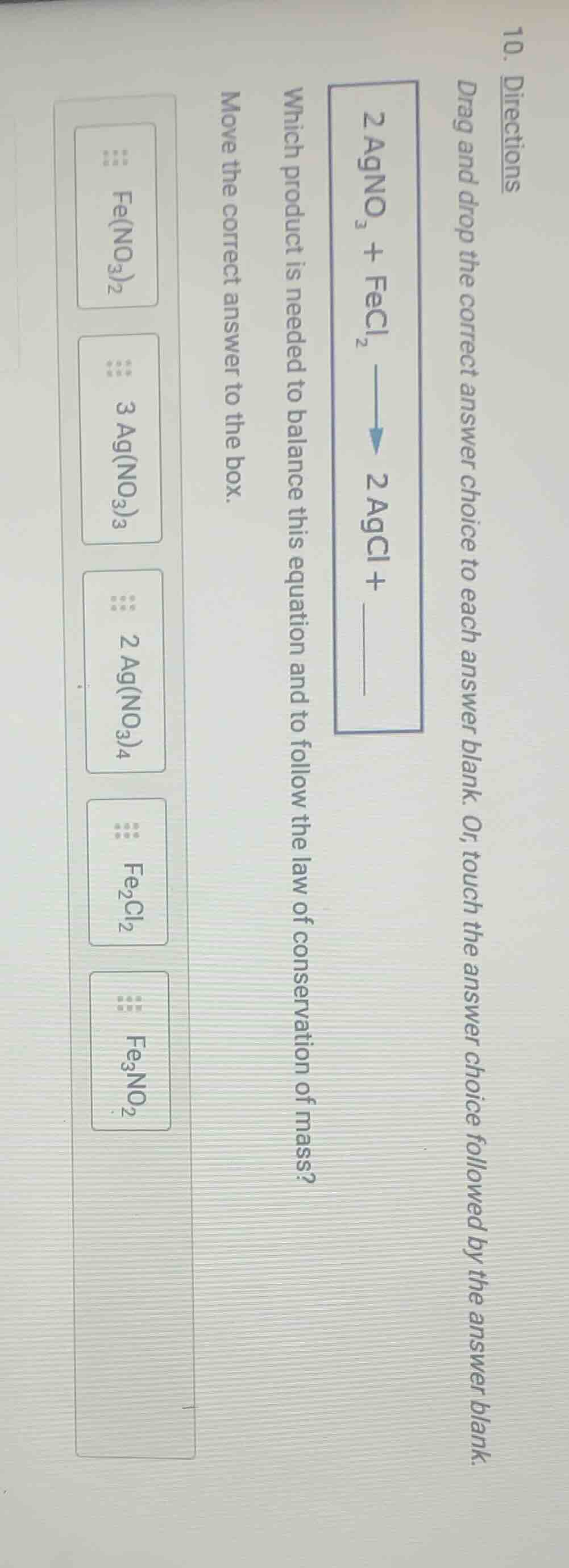
- directions
drag and drop the correct answer choice to each answer blank. or, touch the answer choice followed by the answer blank.
\\(2\text{agno}_3 + \text{fecl}_2 \longrightarrow 2\text{agcl} + \underline{quadquad}\\)
which product is needed to balance this equation and to follow the law of conservation of mass?
move the correct answer to the box.
\text{fe(no}_3\text{)}_2
3\text{ag(no}_3\text{)}_3
2\text{ag(no}_3\text{)}_4
\text{fe}_2\text{cl}_2
\text{fe}_3\text{no}_2
Step1: Count reactant atoms
Reactants: $2\text{AgNO}_3 + \text{FeCl}_2$
- Ag: 2, N: 2, O: 6, Fe: 1, Cl: 2
Step2: Count known product atoms
Known product: $2\text{AgCl}$
- Ag: 2, Cl: 2
Step3: Identify missing atoms
Remaining atoms to form product: Fe:1, N:2, O:6
Step4: Match to product formula
Combine remaining atoms: $\text{Fe(NO}_3\text{)}_2$ (contains 1 Fe, 2 N, 6 O)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\text{Fe(NO}_3\text{)}_2$