QUESTION IMAGE
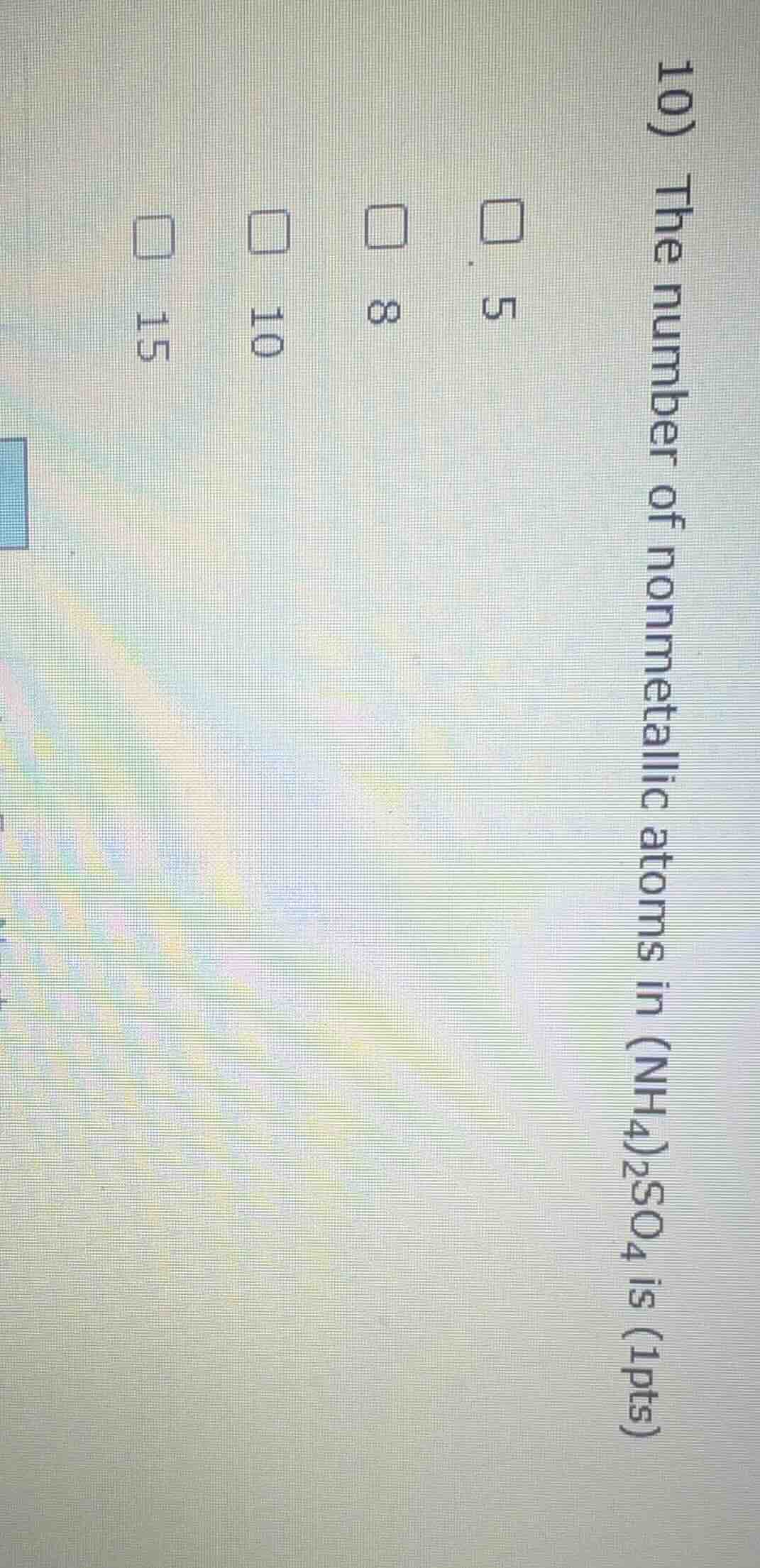
Question
- the number of nonmetallic atoms in (nh4)2so4 is (1pts)
□ 5
□ 8
□ 10
□ 15
Step1: Identify nonmetallic elements
Nonmetals in $\boldsymbol{(NH_4)_2SO_4}$: N, H, S, O
Step2: Count each nonmetal atom
- N: $2 \times 1 = 2$
- H: $2 \times 4 = 8$
- S: $1 \times 1 = 1$
- O: $1 \times 4 = 4$
Step3: Sum all nonmetal atoms
$2 + 8 + 1 + 4 = 15$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{\square}$ 15