QUESTION IMAGE
Question
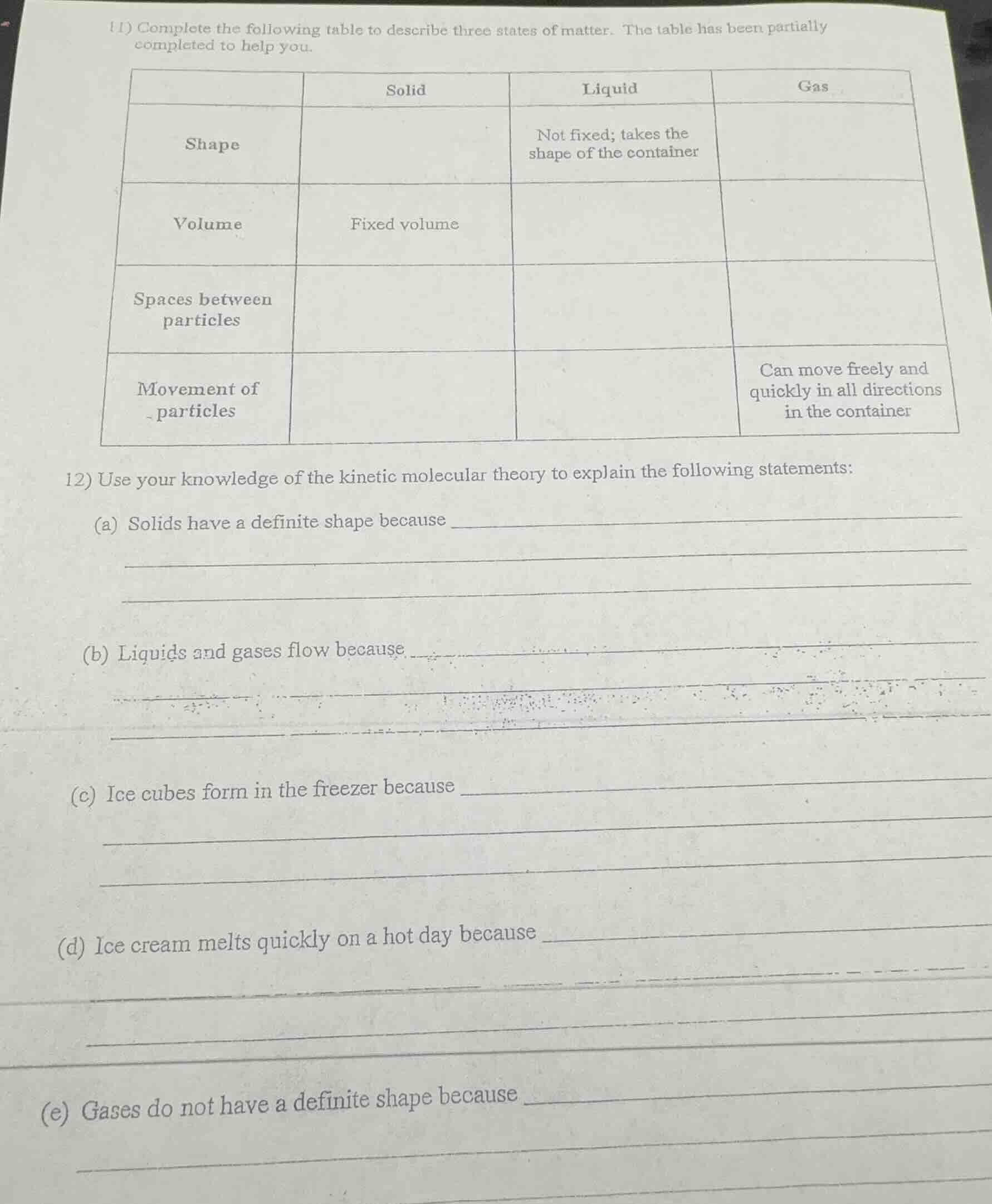
- complete the following table to describe three states of matter. the table has been partially completed to help you.
| solid | liquid | gas | |
|---|---|---|---|
| volume | fixed volume | ||
| spaces between particles | |||
| movement of particles | can move freely and quickly in all directions in the container |
- use your knowledge of the kinetic molecular theory to explain the following statements:
(a) solids have a definite shape because
(b) liquids and gases flow because
(c) ice cubes form in the freezer because
(d) ice cream melts quickly on a hot day because
(e) gases do not have a definite shape because
Question 11: Completing the table for three states of matter
| Solid | Liquid | Gas | |
|---|---|---|---|
| Volume | Fixed volume | Fixed volume | Not fixed (expands to fill container) |
| Spaces between particles | Very small spaces | Moderate spaces (larger than solid) | Very large spaces |
| Movement of particles | Vibrate in fixed positions | Move freely but slowly (slide past each other) | Can move freely and quickly in all directions in the container |
Question 12: Explanations using Kinetic Molecular Theory
(a) Solids have a definite shape because:
The particles in a solid are closely packed and held in fixed positions by strong intermolecular forces. They can only vibrate in place, so the structure of the solid remains rigid and maintains a definite shape.
(b) Liquids and gases flow because:
In liquids, particles are close but can slide past each other (due to weaker intermolecular forces than solids). In gases, particles are far apart and move freely. Both have particles that can move relative to each other, allowing them to flow and take the shape of their container.
(c) Ice cubes form in the freezer because:
When water is cooled in the freezer, its particles lose kinetic energy. As temperature decreases, the particles slow down, and intermolecular forces (like hydrogen bonding in water) cause them to arrange into a fixed, ordered structure (ice), transitioning from liquid to solid (freezing).
(d) Ice cream melts quickly on a hot day because:
On a hot day, heat energy is transferred to the ice cream. This increases the kinetic energy of its particles, overcoming the intermolecular forces holding them in a solid structure. The particles start to move more freely, causing the ice cream to transition from solid to liquid (melting).
(e) Gases do not have a definite shape because:
Gas particles have high kinetic energy and are far apart with weak intermolecular forces. They move freely and rapidly in all directions, spreading out to fill the entire volume of their container, so they take the shape of the container rather than having a fixed shape.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 11: Completing the table for three states of matter
| Solid | Liquid | Gas | |
|---|---|---|---|
| Volume | Fixed volume | Fixed volume | Not fixed (expands to fill container) |
| Spaces between particles | Very small spaces | Moderate spaces (larger than solid) | Very large spaces |
| Movement of particles | Vibrate in fixed positions | Move freely but slowly (slide past each other) | Can move freely and quickly in all directions in the container |
Question 12: Explanations using Kinetic Molecular Theory
(a) Solids have a definite shape because:
The particles in a solid are closely packed and held in fixed positions by strong intermolecular forces. They can only vibrate in place, so the structure of the solid remains rigid and maintains a definite shape.
(b) Liquids and gases flow because:
In liquids, particles are close but can slide past each other (due to weaker intermolecular forces than solids). In gases, particles are far apart and move freely. Both have particles that can move relative to each other, allowing them to flow and take the shape of their container.
(c) Ice cubes form in the freezer because:
When water is cooled in the freezer, its particles lose kinetic energy. As temperature decreases, the particles slow down, and intermolecular forces (like hydrogen bonding in water) cause them to arrange into a fixed, ordered structure (ice), transitioning from liquid to solid (freezing).
(d) Ice cream melts quickly on a hot day because:
On a hot day, heat energy is transferred to the ice cream. This increases the kinetic energy of its particles, overcoming the intermolecular forces holding them in a solid structure. The particles start to move more freely, causing the ice cream to transition from solid to liquid (melting).
(e) Gases do not have a definite shape because:
Gas particles have high kinetic energy and are far apart with weak intermolecular forces. They move freely and rapidly in all directions, spreading out to fill the entire volume of their container, so they take the shape of the container rather than having a fixed shape.