QUESTION IMAGE
Question
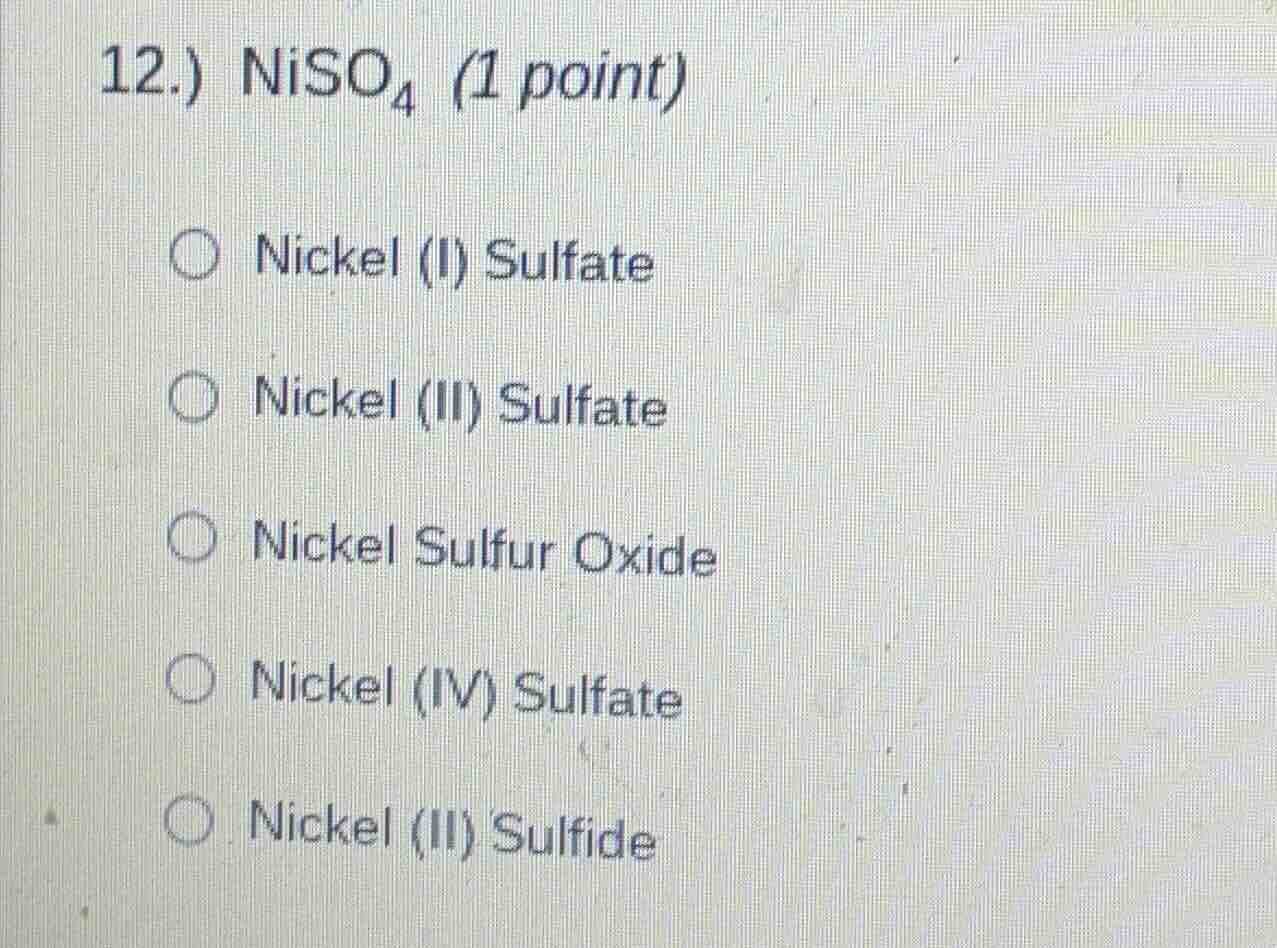
12.) $ce{niso_{4}}$ (1 point)
$\bigcirc$ nickel (i) sulfate
$\bigcirc$ nickel (ii) sulfate
$\bigcirc$ nickel sulfur oxide
$\bigcirc$ nickel (iv) sulfate
$\bigcirc$ nickel (ii) sulfide
Brief Explanations
- Identify the polyatomic ion: $\text{SO}_4^{2-}$ is the sulfate ion, eliminating options with "sulfur oxide" or "sulfide".
- Find the charge of the nickel ion: The compound is neutral, so the nickel charge balances the 2- charge of sulfate. Nickel must have a +2 charge, written as (II) in IUPAC nomenclature.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Nickel (II) Sulfate