QUESTION IMAGE
Question
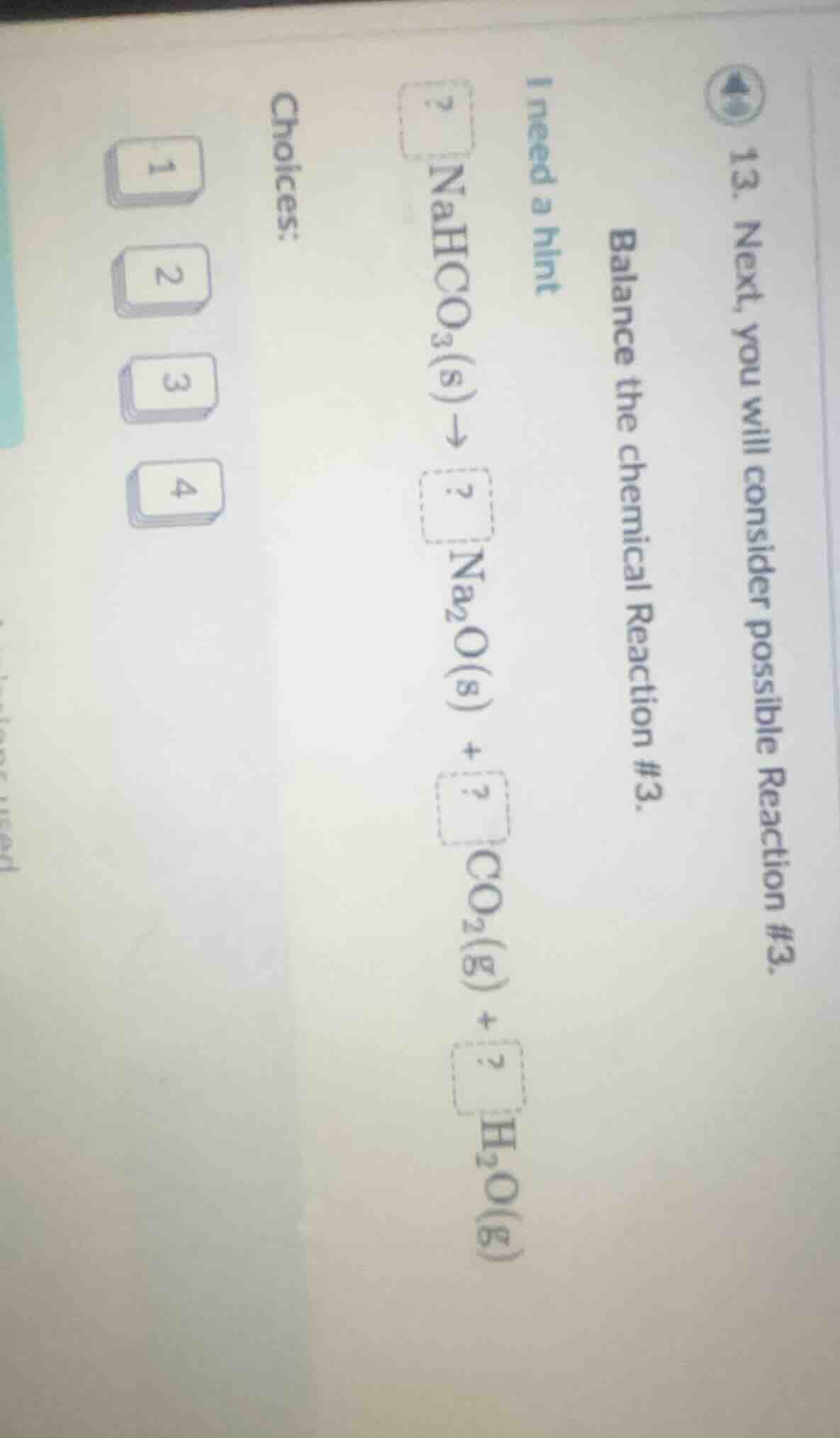
- next, you will consider possible reaction #3.
balance the chemical reaction #3.
i need a hint
$?\\ \ce{nahco3(s)} \
ightarrow ?\\ \ce{na2o(s)} + ?\\ \ce{co2(g)} + ?\\ \ce{h2o(g)}$
choices:
1
2
3
4
Step1: Balance Na atoms
Left: 1 Na in $\text{NaHCO}_3$; Right: 2 Na in $\text{Na}_2\text{O}$. Add coefficient 2 to $\text{NaHCO}_3$:
$2\text{NaHCO}_3(s)
ightarrow ?\text{Na}_2\text{O}(s) + ?\text{CO}_2(g) + ?\text{H}_2\text{O}(g)$
Step2: Balance C atoms
Left: 2 C in $2\text{NaHCO}_3$; Right: 1 C in $\text{CO}_2$. Add coefficient 2 to $\text{CO}_2$:
$2\text{NaHCO}_3(s)
ightarrow ?\text{Na}_2\text{O}(s) + 2\text{CO}_2(g) + ?\text{H}_2\text{O}(g)$
Step3: Balance H atoms
Left: 2 H in $2\text{NaHCO}_3$; Right: 2 H in $\text{H}_2\text{O}$. Add coefficient 1 to $\text{H}_2\text{O}$:
$2\text{NaHCO}_3(s)
ightarrow ?\text{Na}_2\text{O}(s) + 2\text{CO}_2(g) + 1\text{H}_2\text{O}(g)$
Step4: Balance O atoms
Left: $2\times3=6$ O in $2\text{NaHCO}_3$; Right: $1\text{Na}_2\text{O}$ (1 O) + $2\text{CO}_2$ (4 O) + $1\text{H}_2\text{O}$ (1 O) = 6 O. Add coefficient 1 to $\text{Na}_2\text{O}$:
$2\text{NaHCO}_3(s)
ightarrow 1\text{Na}_2\text{O}(s) + 2\text{CO}_2(g) + 1\text{H}_2\text{O}(g)$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The coefficients (in order) are 2, 1, 2, 1.